Basal extrusion drives cell invasion and mechanical stripping of E-cadherin
Preprint posted on 6 November 2018 https://www.biorxiv.org/content/early/2018/11/06/463646
Categories: cancer biology, cell biology
Background
Oncogenesis can start with a mutation to a single cell, resulting in a transformed malignant cell, within a healthy epithelial sheet. It is increasingly apparent that single transformed cells can be detected and squeezed out of the tissue by the surrounding normal neighbours. This process has been observed in flies, cell lines, organoids, zebrafish and in vivo using mouse models (Bielmier et al., 2016; Hogan et al., 2009; Kon et al., 2017; Anton et al., 2018).
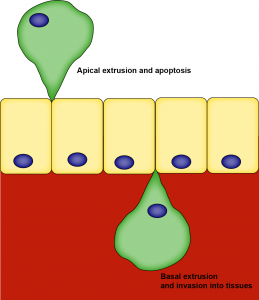
However, what is less clear is the fate of extruded cells. It is currently thought that the direction of extrusion can impact an extruded cells’ fate; if a cell is pushed out apically then it dies or is cleared through the lumen. However, it is unclear what happens to cells that extrude basally.
In this preprint, the authors investigate the fate of KRas-transformed cells surrounded by normal neighbours in zebrafish. Expression of different oncogenes was combined with fluorescent markers to track mutant cells in real-time. Oncogenes which promote extrusion are associated with metastatic cancers so the authors hypothesise that basal extrusion could be a novel mechanism of metastasis.
Key findings of the preprint
KRas-transformed cells can be extruded basally and apically and initiate tumourigenesis.
Firstly, the authors injected EGFP-KRasV12 plasmids driven by the krt4 promoter into one-cell embryos to generate mosaic expression of KRas in epidermal cells. They found that this was sufficient to form cell masses in 58% of zebrafish embryos compared to 1% of EGFP-CAAX controls. Time-lapse imaging of labelled cells over time revealed that KRasV12 cells can extrude both apically and basally, although significantly more basal extrusions were observed. They saw that basal extrusion can lead to invasion and internalisation of mutant cells into different parts of the zebrafish. Although KRas-transformed cells could form cell masses, a large number of mutant cells were lost over time, which could be a result of extrusion observed in time-lapse imaging.
Loss of p53 promotes the survival extruded cells.
During the evolution of cancer, transformed cells accumulate additional mutations which drive tumourigenesis. To model this the authors combined expression of KRasV12 with depletion of p53 via a translation-blocking morpholino. This led to a reduction in basal extrusion of aberrant cells. However, loss of p53 increased the survival of basally invading cells and their ability to survive as internalised masses at later time points.
Extruded cells can spread through the bloodstream and may initiate EMT
After establishing the system, the authors began characterising cells that invade basally. They found that all internalised cells lose the epithelial marker E-cadherin and a small subset expressed the mesenchymal marker N-cadherin. Intriguingly, time lapse imaging in combination with a vasculature reporter was used to identify GFP-KRasV12 cells in the bloodstream with 65% of 42 zebrafish having circulating cells. Finally, using different fluorescent markers for the apical and cytoplasm it was observed that the apical membrane is ripped off during extrusion and the remainder of the cell migrates away. The authors suggest that basal extrusion drives invasion of KRasV12 cells and strips off the apical surfaces and associated E-cadherin.
What I like about this preprint
This preprint demonstrates that extrusion can lead to mutant cells in the circulation of zebrafish, which is a possible new way for oncogenic cells to spread around the body. Although lots of work on the mechanism of extrusion has been carried out, this paper begins to characterise the fate of extruded cells.
The authors looked at how the mechanical force of KrasV12 cells being squeezed out constricts the nucleus and can scrape off the E-cadherin apical membrane proteins. This sudden loss of the apical membrane could have important implications for metastasis and EMT.
I also liked the great videos of cells extruding from zebrafish!
Questions to the authors
What do you think could be regulating whether mutant cells either accumulate into masses or extrude in the different parts of the zebrafish?
In the movies do you ever see KRasV12 cells extravasate from the circulation?
Mechanical stretch can promote YAP translocation into the nucleus, have you looked for YAP nuclear localisation in extruding cells?
References:
Anton, K. A., Kajita, M., Narumi, R., Fujita, Y., & Tada, M. (2018). Src-transformed cells hijack mitosis to extrude from the epithelium. Nature communications, 9(1), 4695.
Bielmeier, C., Alt, S., Weichselberger, V., La Fortezza, M., Harz, H., Jülicher, F., … & Classen, A. K. (2016). Interface contractility between differently fated cells drives cell elimination and cyst formation. Current Biology, 26(5), 563-574.
Hogan, C., Dupré-Crochet, S., Norman, M., Kajita, M., Zimmermann, C., Pelling, A. E., … & Hosoya, H. (2009). Characterization of the interface between normal and transformed epithelial cells. Nature cell biology, 11(4), 460.
Kon, S., Ishibashi, K., Katoh, H., Kitamoto, S., Shirai, T., Tanaka, S., … & Kamasaki, T. (2017). Cell competition with normal epithelial cells promotes apical extrusion of transformed cells through metabolic changes. Nature cell biology, 19(5), 530.
Posted on: 2 January 2019
doi: https://doi.org/10.1242/prelights.6788
Read preprintSign up to customise the site to your preferences and to receive alerts
Register hereAlso in the cancer biology category:
Spatial transcriptomics elucidates medulla niche supporting germinal center response in myasthenia gravis thymoma
Invasion of glioma cells through confined space requires membrane tension regulation and mechano-electrical coupling via Plexin-B2
Modeling transcriptional profiles of gene perturbation with a deep neural network
Also in the cell biology category:
Clusters of lineage-specific genes are anchored by ZNF274 in repressive perinucleolar compartments
Structural basis of respiratory complexes adaptation to cold temperatures
RIPK3 coordinates RHIM domain-dependent inflammatory transcription in neurons
preLists in the cancer biology category:
BSCB-Biochemical Society 2024 Cell Migration meeting
This preList features preprints that were discussed and presented during the BSCB-Biochemical Society 2024 Cell Migration meeting in Birmingham, UK in April 2024. Kindly put together by Sara Morais da Silva, Reviews Editor at Journal of Cell Science.
| List by | Reinier Prosee |
CSHL 87th Symposium: Stem Cells
Preprints mentioned by speakers at the #CSHLsymp23
| List by | Alex Eve |
Journal of Cell Science meeting ‘Imaging Cell Dynamics’
This preList highlights the preprints discussed at the JCS meeting 'Imaging Cell Dynamics'. The meeting was held from 14 - 17 May 2023 in Lisbon, Portugal and was organised by Erika Holzbaur, Jennifer Lippincott-Schwartz, Rob Parton and Michael Way.
| List by | Helen Zenner |
CellBio 2022 – An ASCB/EMBO Meeting
This preLists features preprints that were discussed and presented during the CellBio 2022 meeting in Washington, DC in December 2022.
| List by | Nadja Hümpfer et al. |
Fibroblasts
The advances in fibroblast biology preList explores the recent discoveries and preprints of the fibroblast world. Get ready to immerse yourself with this list created for fibroblasts aficionados and lovers, and beyond. Here, my goal is to include preprints of fibroblast biology, heterogeneity, fate, extracellular matrix, behavior, topography, single-cell atlases, spatial transcriptomics, and their matrix!
| List by | Osvaldo Contreras |
Single Cell Biology 2020
A list of preprints mentioned at the Wellcome Genome Campus Single Cell Biology 2020 meeting.
| List by | Alex Eve |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
Lung Disease and Regeneration
This preprint list compiles highlights from the field of lung biology.
| List by | Rob Hynds |
Anticancer agents: Discovery and clinical use
Preprints that describe the discovery of anticancer agents and their clinical use. Includes both small molecules and macromolecules like biologics.
| List by | Zhang-He Goh |
Biophysical Society Annual Meeting 2019
Few of the preprints that were discussed in the recent BPS annual meeting at Baltimore, USA
| List by | Joseph Jose Thottacherry |
Also in the cell biology category:
BSCB-Biochemical Society 2024 Cell Migration meeting
This preList features preprints that were discussed and presented during the BSCB-Biochemical Society 2024 Cell Migration meeting in Birmingham, UK in April 2024. Kindly put together by Sara Morais da Silva, Reviews Editor at Journal of Cell Science.
| List by | Reinier Prosee |
‘In preprints’ from Development 2022-2023
A list of the preprints featured in Development's 'In preprints' articles between 2022-2023
| List by | Alex Eve, Katherine Brown |
preLights peer support – preprints of interest
This is a preprint repository to organise the preprints and preLights covered through the 'preLights peer support' initiative.
| List by | preLights peer support |
The Society for Developmental Biology 82nd Annual Meeting
This preList is made up of the preprints discussed during the Society for Developmental Biology 82nd Annual Meeting that took place in Chicago in July 2023.
| List by | Joyce Yu, Katherine Brown |
CSHL 87th Symposium: Stem Cells
Preprints mentioned by speakers at the #CSHLsymp23
| List by | Alex Eve |
Journal of Cell Science meeting ‘Imaging Cell Dynamics’
This preList highlights the preprints discussed at the JCS meeting 'Imaging Cell Dynamics'. The meeting was held from 14 - 17 May 2023 in Lisbon, Portugal and was organised by Erika Holzbaur, Jennifer Lippincott-Schwartz, Rob Parton and Michael Way.
| List by | Helen Zenner |
9th International Symposium on the Biology of Vertebrate Sex Determination
This preList contains preprints discussed during the 9th International Symposium on the Biology of Vertebrate Sex Determination. This conference was held in Kona, Hawaii from April 17th to 21st 2023.
| List by | Martin Estermann |
Alumni picks – preLights 5th Birthday
This preList contains preprints that were picked and highlighted by preLights Alumni - an initiative that was set up to mark preLights 5th birthday. More entries will follow throughout February and March 2023.
| List by | Sergio Menchero et al. |
CellBio 2022 – An ASCB/EMBO Meeting
This preLists features preprints that were discussed and presented during the CellBio 2022 meeting in Washington, DC in December 2022.
| List by | Nadja Hümpfer et al. |
Fibroblasts
The advances in fibroblast biology preList explores the recent discoveries and preprints of the fibroblast world. Get ready to immerse yourself with this list created for fibroblasts aficionados and lovers, and beyond. Here, my goal is to include preprints of fibroblast biology, heterogeneity, fate, extracellular matrix, behavior, topography, single-cell atlases, spatial transcriptomics, and their matrix!
| List by | Osvaldo Contreras |
EMBL Synthetic Morphogenesis: From Gene Circuits to Tissue Architecture (2021)
A list of preprints mentioned at the #EESmorphoG virtual meeting in 2021.
| List by | Alex Eve |
FENS 2020
A collection of preprints presented during the virtual meeting of the Federation of European Neuroscience Societies (FENS) in 2020
| List by | Ana Dorrego-Rivas |
Planar Cell Polarity – PCP
This preList contains preprints about the latest findings on Planar Cell Polarity (PCP) in various model organisms at the molecular, cellular and tissue levels.
| List by | Ana Dorrego-Rivas |
BioMalPar XVI: Biology and Pathology of the Malaria Parasite
[under construction] Preprints presented at the (fully virtual) EMBL BioMalPar XVI, 17-18 May 2020 #emblmalaria
| List by | Dey Lab, Samantha Seah |
1
Cell Polarity
Recent research from the field of cell polarity is summarized in this list of preprints. It comprises of studies focusing on various forms of cell polarity ranging from epithelial polarity, planar cell polarity to front-to-rear polarity.
| List by | Yamini Ravichandran |
TAGC 2020
Preprints recently presented at the virtual Allied Genetics Conference, April 22-26, 2020. #TAGC20
| List by | Maiko Kitaoka et al. |
3D Gastruloids
A curated list of preprints related to Gastruloids (in vitro models of early development obtained by 3D aggregation of embryonic cells). Updated until July 2021.
| List by | Paul Gerald L. Sanchez and Stefano Vianello |
ECFG15 – Fungal biology
Preprints presented at 15th European Conference on Fungal Genetics 17-20 February 2020 Rome
| List by | Hiral Shah |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
EMBL Seeing is Believing – Imaging the Molecular Processes of Life
Preprints discussed at the 2019 edition of Seeing is Believing, at EMBL Heidelberg from the 9th-12th October 2019
| List by | Dey Lab |
Autophagy
Preprints on autophagy and lysosomal degradation and its role in neurodegeneration and disease. Includes molecular mechanisms, upstream signalling and regulation as well as studies on pharmaceutical interventions to upregulate the process.
| List by | Sandra Malmgren Hill |
Lung Disease and Regeneration
This preprint list compiles highlights from the field of lung biology.
| List by | Rob Hynds |
Cellular metabolism
A curated list of preprints related to cellular metabolism at Biorxiv by Pablo Ranea Robles from the Prelights community. Special interest on lipid metabolism, peroxisomes and mitochondria.
| List by | Pablo Ranea Robles |
BSCB/BSDB Annual Meeting 2019
Preprints presented at the BSCB/BSDB Annual Meeting 2019
| List by | Dey Lab |
MitoList
This list of preprints is focused on work expanding our knowledge on mitochondria in any organism, tissue or cell type, from the normal biology to the pathology.
| List by | Sandra Franco Iborra |
ASCB/EMBO Annual Meeting 2018
This list relates to preprints that were discussed at the recent ASCB conference.
| List by | Dey Lab, Amanda Haage |











 (No Ratings Yet)
(No Ratings Yet)