Clathrin plaques form mechanotransducing platforms
Preprint posted on 14 May 2018 https://www.biorxiv.org/content/early/2018/05/14/321885?%3Fcollection=
Clathrin plaques are the new focal adhesions: Adhesive clathrin plaque association with the cytoskeleton provides a novel platform for mechanosensing, including the regulation of YAP/TAZ signaling.
Selected by Amanda HaageCategories: biophysics, cell biology
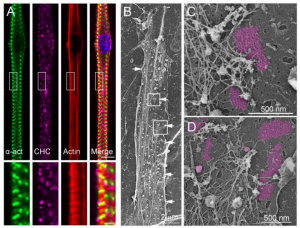
Why This Is Cool – The authors provide evidence that two relatively new discoveries in cell biology, mechanotransduction via YAP/TAZ signaling and large clathrin plaque structures on the plasma membrane, work together to produce a novel platform for sensing mechanical input, connecting and regulating the cytoskeleton, and integrating various signaling pathways. These are the roles that have previously been established for focal adhesions. It makes sense that cells could complete these essential tasks in a variety of ways using other large membrane-associated protein complexes. The authors provide exhaustive evidence for the mechanisms and relevance of this new system. First, they demonstrate using beautiful images of immunogold labeling with metal-replica EM (Fig1) the precise organization of the clathrin plaques surrounded by branched actin and desmin intermediate filaments on primary mouse myotubes. Next they show that these structures respond to mechanical stimuli by subjecting the myotubes to cyclic stretching. Upon stretching the plaques decrease in size supposedly due to an increase in endocytosis and the YAP/TAZ mechanotransducers canonically accumulate in the nucleus with a concurrent increase in their target genes’ expression levels. Interestingly, myotubes without clathrin plaques had high levels of YAP/TAZ nuclear staining without stretching with no obvious response to stretch. The authors go on to demonstrate this is because the clathrin plaques act as sticky nets that sequester YAP/TAZ until the cell receives a mechanical input. They propose that YAP/TAZ gets stuck at the actin network surrounding clathrin plaques largely through an interaction between TAZ and Dynamin 2. Myotubes without Dynamin 2 lose their actin organization, and their ability to translocate YAP/TAZ in response to stretch. Now that they have established a mechanism for how clathrin plaques can act as mechanosensors, the authors go on to show the relevance of this mechanism in vivo. A type of centronuclear myopathy is caused by mutations in Dynamin 2. By using a knock-in mouse model for the most common human mutation linked to this disease, they demonstrate similar phenotypes to the Dynamin 2 knockout myotubes. These mice were found to have disorganized clathrin plaques, TAZ, and desmin in their muscles. In addition, primary culture of their mytotubes revealed a decrease in TAZ nuclear localization without mechanical input. To really drive the point home, the authors also repeat these findings in immortalized myotubes from a centronuclear myopathy human patient.
Why I Selected It – The idea that cells can sense and respond to the physical properties of their microenvironment has always fascinated me. It’s a newer concept that has burst into a huge field of cell biology. We are now getting beyond the initial discoveries of this phenomenon and learning just how much it permeates every part of cell biology. Mechanotransduction is not just a field for cell migration or extracellular matrix people, but is a field for everyone.
Open Questions –
- Has YAP/TAZ translocation via endocytosis been directly observed? Is it something that could be observed via live imaging?
- Do the clathrin plaques ever recover in size and YAP/TAZ content after extended periods of relaxation?
- Could the Dynamin 2 – TAZ interaction be a realistic drug target for centronuclear myopathy?
Related References –
- What is YAP/TAZ?
- Dupont S. Role of YAP/TAZ in cell-matrix adhesion-mediates signaling and mechanotransduction. Exp Cell Res. (2016) 10;343(1):42-53/
- What are clathrin plaques?
- Lampe M., Vassilopoulos S., Merrifield C. Clathrin coated pits, plaques and adhesion. J Struct Biol. (2016) 196(1):48-56.
- Mutations in Dynamin 2 cause centronuclear myopathy
- Bitoun M., Maugenre S., Jeannet PY., Lacene E., Ferrer X., Laforet P., Martin JJ., Laporte J., Lochmuller H., Beggs AH., Fardeau M., Eymard B., Romero NB., Guicheney P. Mutations in dynamin 2 cause dominant centronuclear myopathy. Nat Genet. (2005) 37(11):1207-9.
Posted on: 6 June 2018
Read preprintSign up to customise the site to your preferences and to receive alerts
Register hereAlso in the biophysics category:
Structural basis of respiratory complexes adaptation to cold temperatures
Actin polymerization drives lumen formation in a human epiblast model
Learning a conserved mechanism for early neuroectoderm morphogenesis
Also in the cell biology category:
Fetal brain response to maternal inflammation requires microglia
Alteration of long and short-term hematopoietic stem cell ratio causes myeloid-biased hematopoiesis
Clusters of lineage-specific genes are anchored by ZNF274 in repressive perinucleolar compartments
preLists in the biophysics category:
preLights peer support – preprints of interest
This is a preprint repository to organise the preprints and preLights covered through the 'preLights peer support' initiative.
| List by | preLights peer support |
66th Biophysical Society Annual Meeting, 2022
Preprints presented at the 66th BPS Annual Meeting, Feb 19 - 23, 2022 (The below list is not exhaustive and the preprints are listed in no particular order.)
| List by | Soni Mohapatra |
EMBL Synthetic Morphogenesis: From Gene Circuits to Tissue Architecture (2021)
A list of preprints mentioned at the #EESmorphoG virtual meeting in 2021.
| List by | Alex Eve |
Biophysical Society Meeting 2020
Some preprints presented at the Biophysical Society Meeting 2020 in San Diego, USA.
| List by | Tessa Sinnige |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
EMBL Seeing is Believing – Imaging the Molecular Processes of Life
Preprints discussed at the 2019 edition of Seeing is Believing, at EMBL Heidelberg from the 9th-12th October 2019
| List by | Dey Lab |
Biomolecular NMR
Preprints related to the application and development of biomolecular NMR spectroscopy
| List by | Reid Alderson |
Biophysical Society Annual Meeting 2019
Few of the preprints that were discussed in the recent BPS annual meeting at Baltimore, USA
| List by | Joseph Jose Thottacherry |
Also in the cell biology category:
BSCB-Biochemical Society 2024 Cell Migration meeting
This preList features preprints that were discussed and presented during the BSCB-Biochemical Society 2024 Cell Migration meeting in Birmingham, UK in April 2024. Kindly put together by Sara Morais da Silva, Reviews Editor at Journal of Cell Science.
| List by | Reinier Prosee |
‘In preprints’ from Development 2022-2023
A list of the preprints featured in Development's 'In preprints' articles between 2022-2023
| List by | Alex Eve, Katherine Brown |
preLights peer support – preprints of interest
This is a preprint repository to organise the preprints and preLights covered through the 'preLights peer support' initiative.
| List by | preLights peer support |
The Society for Developmental Biology 82nd Annual Meeting
This preList is made up of the preprints discussed during the Society for Developmental Biology 82nd Annual Meeting that took place in Chicago in July 2023.
| List by | Joyce Yu, Katherine Brown |
CSHL 87th Symposium: Stem Cells
Preprints mentioned by speakers at the #CSHLsymp23
| List by | Alex Eve |
Journal of Cell Science meeting ‘Imaging Cell Dynamics’
This preList highlights the preprints discussed at the JCS meeting 'Imaging Cell Dynamics'. The meeting was held from 14 - 17 May 2023 in Lisbon, Portugal and was organised by Erika Holzbaur, Jennifer Lippincott-Schwartz, Rob Parton and Michael Way.
| List by | Helen Zenner |
9th International Symposium on the Biology of Vertebrate Sex Determination
This preList contains preprints discussed during the 9th International Symposium on the Biology of Vertebrate Sex Determination. This conference was held in Kona, Hawaii from April 17th to 21st 2023.
| List by | Martin Estermann |
Alumni picks – preLights 5th Birthday
This preList contains preprints that were picked and highlighted by preLights Alumni - an initiative that was set up to mark preLights 5th birthday. More entries will follow throughout February and March 2023.
| List by | Sergio Menchero et al. |
CellBio 2022 – An ASCB/EMBO Meeting
This preLists features preprints that were discussed and presented during the CellBio 2022 meeting in Washington, DC in December 2022.
| List by | Nadja Hümpfer et al. |
Fibroblasts
The advances in fibroblast biology preList explores the recent discoveries and preprints of the fibroblast world. Get ready to immerse yourself with this list created for fibroblasts aficionados and lovers, and beyond. Here, my goal is to include preprints of fibroblast biology, heterogeneity, fate, extracellular matrix, behavior, topography, single-cell atlases, spatial transcriptomics, and their matrix!
| List by | Osvaldo Contreras |
EMBL Synthetic Morphogenesis: From Gene Circuits to Tissue Architecture (2021)
A list of preprints mentioned at the #EESmorphoG virtual meeting in 2021.
| List by | Alex Eve |
FENS 2020
A collection of preprints presented during the virtual meeting of the Federation of European Neuroscience Societies (FENS) in 2020
| List by | Ana Dorrego-Rivas |
Planar Cell Polarity – PCP
This preList contains preprints about the latest findings on Planar Cell Polarity (PCP) in various model organisms at the molecular, cellular and tissue levels.
| List by | Ana Dorrego-Rivas |
BioMalPar XVI: Biology and Pathology of the Malaria Parasite
[under construction] Preprints presented at the (fully virtual) EMBL BioMalPar XVI, 17-18 May 2020 #emblmalaria
| List by | Dey Lab, Samantha Seah |
1
Cell Polarity
Recent research from the field of cell polarity is summarized in this list of preprints. It comprises of studies focusing on various forms of cell polarity ranging from epithelial polarity, planar cell polarity to front-to-rear polarity.
| List by | Yamini Ravichandran |
TAGC 2020
Preprints recently presented at the virtual Allied Genetics Conference, April 22-26, 2020. #TAGC20
| List by | Maiko Kitaoka et al. |
3D Gastruloids
A curated list of preprints related to Gastruloids (in vitro models of early development obtained by 3D aggregation of embryonic cells). Updated until July 2021.
| List by | Paul Gerald L. Sanchez and Stefano Vianello |
ECFG15 – Fungal biology
Preprints presented at 15th European Conference on Fungal Genetics 17-20 February 2020 Rome
| List by | Hiral Shah |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
EMBL Seeing is Believing – Imaging the Molecular Processes of Life
Preprints discussed at the 2019 edition of Seeing is Believing, at EMBL Heidelberg from the 9th-12th October 2019
| List by | Dey Lab |
Autophagy
Preprints on autophagy and lysosomal degradation and its role in neurodegeneration and disease. Includes molecular mechanisms, upstream signalling and regulation as well as studies on pharmaceutical interventions to upregulate the process.
| List by | Sandra Malmgren Hill |
Lung Disease and Regeneration
This preprint list compiles highlights from the field of lung biology.
| List by | Rob Hynds |
Cellular metabolism
A curated list of preprints related to cellular metabolism at Biorxiv by Pablo Ranea Robles from the Prelights community. Special interest on lipid metabolism, peroxisomes and mitochondria.
| List by | Pablo Ranea Robles |
BSCB/BSDB Annual Meeting 2019
Preprints presented at the BSCB/BSDB Annual Meeting 2019
| List by | Dey Lab |
MitoList
This list of preprints is focused on work expanding our knowledge on mitochondria in any organism, tissue or cell type, from the normal biology to the pathology.
| List by | Sandra Franco Iborra |
ASCB/EMBO Annual Meeting 2018
This list relates to preprints that were discussed at the recent ASCB conference.
| List by | Dey Lab, Amanda Haage |











 (No Ratings Yet)
(No Ratings Yet)