Severe immunodeficiency in mice lacking DNA repair factors XLF and Mri
Posted on: 27 July 2020
Preprint posted on 2 July 2020
Article now published in Aging at http://dx.doi.org/10.18632/aging.202346
Stop SCID-ing around! The role of NHEJ factors XLF and MRI in mouse immune system maturation
Selected by Jennifer Ann BlackCategories: immunology
Background:
DNA double strain breaks (DSBs) are deleterious lesions that can seriously compromise genome stability. However, under certain circumstances, the repair of DSBs can generate diversity. For example, during T and B cell (lymphocytes) development(1). These cells undergo a process called V(D)J recombination to produce a vast diversity of antibodies and T cell receptors (TCRs) available for an immune response. Here, DSB repair is facilitated by non-homologous end joining (NHEJ). Several key enzymes act during NHEJ. Initially, the heterodimeric complex (composed of Ku80 and Ku70) is recruited to the DSB, followed by the DNA repair kinase DNA-PK catalytic subunit (DNA-PKcs) which can signal for downstream factors to be recruited including XRCC4-like (XLF; involved in the ligation of DNA ends) and factors which stabilise the NHEJ complex; PAXX (paralog of XRCC4 and XLF) and Mri (modulator of retroviral infection; 2). In mice, NHEJ deficiency is linked to severe combined immunodeficiency (SCID; 3&4), however when either XLF, PAXX or Mri are inactivated, mild immunodeficiency only occurs in XLF deficient mice but not for Mri or PAXX deletion suggesting loss of XLF can be compensated for (i.e possesses functional redundancy). Functional redundancy can occur when factors have very similar or identical functions or their functions complement. When XLF is deleted in combination with either Mri or PAXX, deficient embryos don’t survive indicating some interplay between these factors during immune system development (and other, wider roles). Here, the authors generate several double deficient mouse strains, by reducing or inactivating Trp53 activity (a tumour suppressor protein; 5), which prevents embryonic lethality. They use these strains to understand the roles of these factors in B and T cell development.
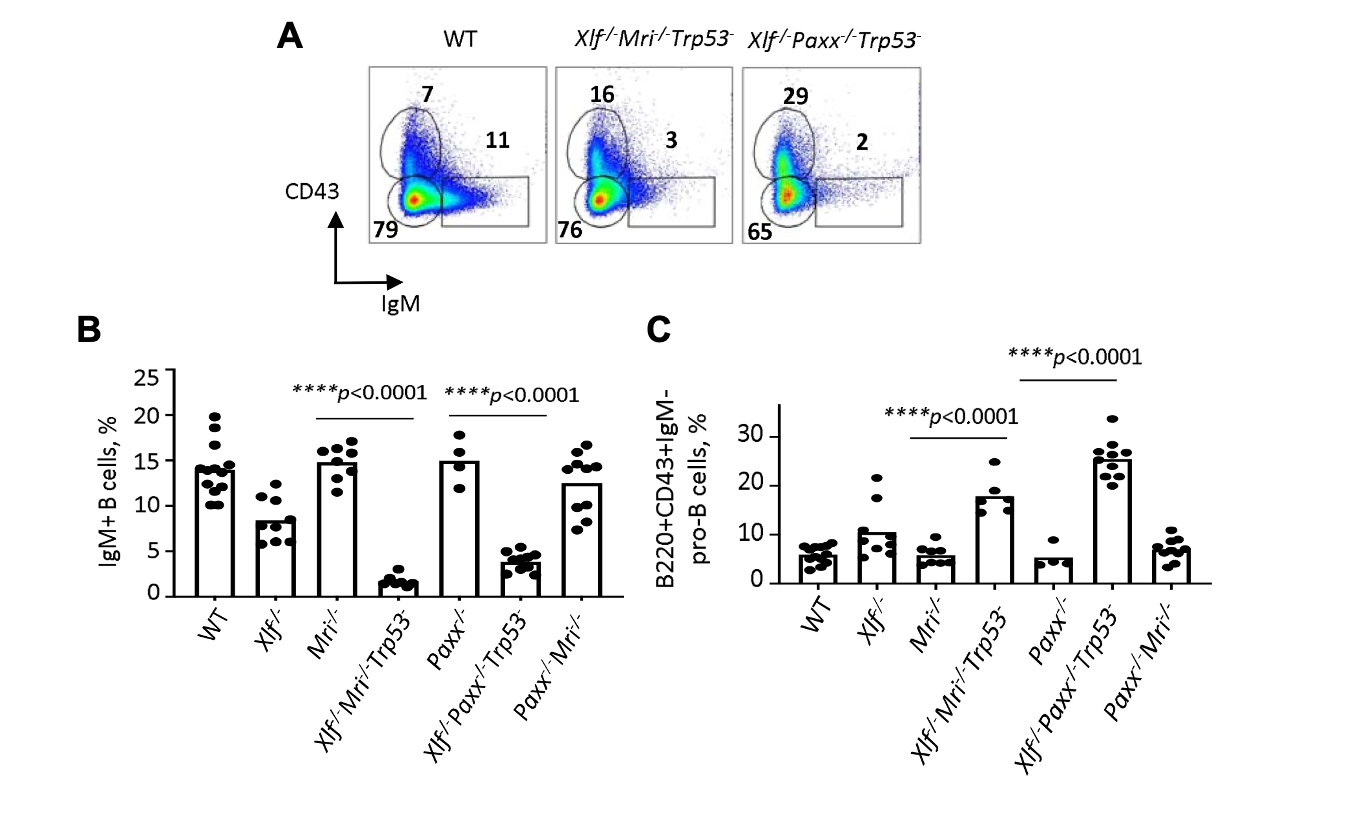
Figure shows (A) FACs plots showing B-cell development in WT mice and double deficient mice (control mouse FACs plots are available in Figure 3A of the preprint). (B) Graph shows the percentage of IgM+ B-cells in WT mice and KO mice. (C) Graph shows the percentage of progenitor B-cells (B220+CD43+IgM-) in WT mice and KO mice. Statistics were generated using a one-way ANOVA test. Figure was adapted from Figure 3 (version 2 of preprint) under a CC-BY-NC-ND 4.0 International license.
Key Findings:
- Combined deletion of XLF with either Mri or PAXX can be achieved by inactivating Trp53 activity
Prior work from the authors revealed that the combined deletion of XLF+PAXX in mice, which results in embryonic lethality, was linked to Trp53 activation (6). Using this information, the authors generate XLF-/-Mri-/-Trp53+/- and XLF-/-PAXX-/-Trp53+/- mice (which they confirmed by PCR analysis) via several rounds of crossing between single deficient mice and mice heterozygote for Trp53+/-. By attenuating Trp53 activity, these two strains of double KO mice can survive into adulthood though Mri-/-XLF-/-Trp53+/- mice were notably lighter (by ~7g) than comparable single KO mice.
- Mice lacking XLF and either Mri or PAXX are immunodeficient
Next, the authors examined how the combined loss of XLF+Mri or XLF+PAXX affected B and T cell development. By harvesting the spleen and the thymus from these deficient mice and appropriate control strains, the authors detected changes in the number of splenocytes and thymocytes. Mri-/-XLF-/-Trp53+/- and XLF-/-PAXX-/-Trp53+/- mice had less splenocytes (~20 fold for CD19+ B cells for XLF+Mri and up to 600 fold for CD19+ B cells for XLF+PAXX) and less thymocytes (9-fold for XLF+Mri and ~40 fold for XLF+PAXX). In keeping with these observations, the authors noted a reduced spleen development relative to the controls, particularly in the XLF-/-PAXX-/-Trp53+/- mice. In all, Mri-/-XLF-/-Trp53+/- and XLF-/-PAXX-/-Trp53+/- mice show immunodeficiencies (they have a SCID like phenotype with the authors refer to as “leaky SCID”) and a level of functional redundancy exists between XLF and PAXX and XLF and Mri. Their data also suggests that the B cells being made in the bone marrow are compromised before making it to the spleen.
- Deletion of XLF+Mri or XLF+PAXX compromises B cell maturation
Next, the authors used flow cytometry to investigate B cells from the bone marrow (from the femora of mice). Here, they reveal that the deletion of XLF+Mri or XLF+PAXX resulted in higher numbers (2-3 fold and 3-4 fold respectively) of progenitor B cells (marked by B220+CD43+IgM-) and lower levels of immature B cells (IgM+ cells) indicating the progenitor B cells generated by these mice are largely defective during their maturation into immature B cells.
- Deleting PAXX and Mri does not cause immunodeficiency in mice but deleting Mri and DNA-PKcs is embryonic lethal
When PAXX or Mri are deleted in combination with XLF, this effect is embryonic lethal, or, when Trp53 is inactivated, their deletion compromises B cell development. With this in mind, the authors next asked what happens if both PAXX and Mri are deleted in mice. In all, the found no clear differences in PAXX-/-Mri-/- mice (which were viable and fertile) compared with appropriate controls suggesting these factors do not genetically interact. However, when Mri was deleted in combination with the kinase DNA-PKcs, viable young could not be recovered. Therefore, they concluded that Mri and DNA-PKcs do interact genetically.
What I liked about this preprint:
DNA repair mechanics, immune system development and diversity are very complex yet intertwined biological processes. Here, the authors built upon their previous findings to ask questions about the interplay between DNA repair factors (required for NHEJ) and immune system diversity and development using deficient mice strains. Their findings showing functional redundancy between XLF, Mri and PAXX in addition to the mouse strains generated will aid future research into this area.
Questions for the authors:
Q1: NHEJ is an important and the primary pathway selected for repair of DSBs in mammalian organisms. It is possible that over time, DSBs will be repaired poorly or un-repaired if NHEJ is compromised. Do your XLF-/-Mri-/-Trp53+/- and XLF-/-PAXX-/-Trp53+/- mice show any longer-term effects as adults? For example, tumours, sensitivity to genotoxins or premature aging?
Q2: Here, you have looked mainly at B cell development. As you mention, T cell development is more challenging to examine. Do you plan to use your mice to investigate T cell and TCR development? What are some of the challenges of investigating T cell development?
Q3: Your data shows that the maturation of B cells (from pro-B to pre-B) is negatively affected. DSBs can also occur in pro-B cells to help with IgH chain assembly leading to pre-B cells meaning the pro-Bs can also be affected by the loss of NHEJ. Given this, despite the mice producing more pro-B cells, do you notice a higher rate of apoptosis or defects in these pro-B cells?
References:
- Roth, D.B. V(D)J Recombination: mechanisms, errors, and fidelity. Microbiol. Spectr 2 (2016).
- Chang, H.H.Y., Pannunzio, N.R., Adachi, N., and Lieber, M.R. Non-homologous DNA end joining and alternative pathways to double-strand break repair. Nat. Rev. Mol. Cell. Bio 18 (2017).
- Zhu, C,. Bogue, M.A,. Lim, D.S,. et al. Ku86-deficient mice exhibit severe combined immunodeficiency and defective processing of V(D)J recombination intermediates. Cell 86 (1996).
- Taccioli, G.E,. Amatucci, A.G,. Beamish, H.J,. et al. Targeted disruption of the catalytic subunit of the DNA-PK gene in mice confers severe combined immunodeficiency and radiosensitivity. Immunity 9 (1998).
- Mantovani, F., Collavin, L., and Del Sal, G. Mutant p53 as a guardian of the cancer cell. Cell Death & Diff. 26 (2019).
- Castaneda-Zegarra, S., Xing, M., Gago-Fuentes, R., Saeterstad, S., and Oksenych, V. Synthetic lethality between DNA repair factors XLF and PAXX is rescued by inactivation of Trp53. DNA Repair 73 (2019).
doi: https://doi.org/10.1242/prelights.23473
Read preprintSign up to customise the site to your preferences and to receive alerts
Register hereAlso in the immunology category:
Gut microbiome changes over the course of multiple sclerosis differentially influence autoimmune neuroinflammation
Carole Djagang et al.
Human Assembloid Model of Emergent Neurotropic Enteroviruses
Mitchell Sarmie
Small Molecule Agonists of TREM2 Reprogram Microglia and Protect Synapses in Human Alzheimer’s Models
Dina Kabbara
preLists in the immunology category:
BSDB Spring Meeting: Molecules to Morphogenesis
The British Society for Developmental Biology (BSDB) Spring Meeting Molecules to Morphogenesis was held from 23–26 March 2026 at the University of Warwick (UK). This meeting brought together a vibrant community of researchers to discuss how molecular mechanisms are integrated across scales to drive morphogenesis, spanning diverse model systems and approaches. This preList contains preprints by presenters from the talk and poster sessions at the meeting. Please do get in touch at preLights@biologists.com if you notice any relevant preprints that we may have missed.
| List by | Ingrid Tsang |
SciELO preprints – From 2025 onwards
SciELO has become a cornerstone of open, multilingual scholarly communication across Latin America. Its preprint server, SciELO preprints, is expanding the global reach of preprinted research from the region (for more information, see our interview with Carolina Tanigushi). This preList brings together biological, English language SciELO preprints to help readers discover emerging work from the Global South. By highlighting these preprints in one place, we aim to support visibility, encourage early feedback, and showcase the vibrant research communities contributing to SciELO’s open science ecosystem.
| List by | Carolina Tanigushi |
Community-driven preList – Immunology
In this community-driven preList, a group of preLighters, with expertise in different areas of immunology have worked together to create this preprint reading list.
| List by | Felipe Del Valle Batalla et al. |
Journal of Cell Science meeting ‘Imaging Cell Dynamics’
This preList highlights the preprints discussed at the JCS meeting 'Imaging Cell Dynamics'. The meeting was held from 14 - 17 May 2023 in Lisbon, Portugal and was organised by Erika Holzbaur, Jennifer Lippincott-Schwartz, Rob Parton and Michael Way.
| List by | Helen Zenner |
Fibroblasts
The advances in fibroblast biology preList explores the recent discoveries and preprints of the fibroblast world. Get ready to immerse yourself with this list created for fibroblasts aficionados and lovers, and beyond. Here, my goal is to include preprints of fibroblast biology, heterogeneity, fate, extracellular matrix, behavior, topography, single-cell atlases, spatial transcriptomics, and their matrix!
| List by | Osvaldo Contreras |
Single Cell Biology 2020
A list of preprints mentioned at the Wellcome Genome Campus Single Cell Biology 2020 meeting.
| List by | Alex Eve |
Autophagy
Preprints on autophagy and lysosomal degradation and its role in neurodegeneration and disease. Includes molecular mechanisms, upstream signalling and regulation as well as studies on pharmaceutical interventions to upregulate the process.
| List by | Sandra Malmgren Hill |
Antimicrobials: Discovery, clinical use, and development of resistance
Preprints that describe the discovery of new antimicrobials and any improvements made regarding their clinical use. Includes preprints that detail the factors affecting antimicrobial selection and the development of antimicrobial resistance.
| List by | Zhang-He Goh |
Zebrafish immunology
A compilation of cutting-edge research that uses the zebrafish as a model system to elucidate novel immunological mechanisms in health and disease.
| List by | Shikha Nayar |











 (No Ratings Yet)
(No Ratings Yet)