SLC19A1 is a cyclic dinucleotide transporter
Posted on: 13 February 2019 , updated on: 14 February 2019
Preprint posted on 4 February 2019
SLC19A1 is an importer of the immunotransmitter cGAMP
Posted on: , updated on: 14 February 2019
Preprint posted on 3 February 2019
Article now published in Molecular Cell at http://dx.doi.org/10.1016/j.molcel.2019.05.006
2'3'-cGAMP is an immunotransmitter produced by cancer cells and regulated by ENPP1
Posted on: , updated on: 14 February 2019
Preprint posted on 3 February 2019
Article now published in Nature Cancer at http://dx.doi.org/10.1038/s43018-020-0028-4
Export and import controls on cyclic dinucleotides in immunity – Three preprints shed light on the immunotransmitter cGAMP
Selected by Connor RosenCategories: immunology
Background:
Cytosolic DNA is a major signal to trigger immune activation and anti-viral immunity. One pathway leading to immune activation downstream of cytosolic DNA sensing in mammals is the cGAS-STING pathway. cGAS binds to cytosolic double-stranded DNA and synthesizes the second messenger 2’3’-cyclic-GMP-AMP (cGAMP), which activates STING, eventually leading to type I interferon production, innate immune activation, and a variety of anti-viral effects. STING agonists, including synthetic cyclic-dinucleotides (CDNs) are additionally under clinical investigation as novel cancer immunotherapies, owing to their ability to stimulate favorable anti-tumor responses in mouse models. Recent data also indicate that there may be a role for extracellular cGAMP as a diffusible second messenger, particularly in cancer models – however, the mechanisms by which cGAMP crosses the plasma membrane to trigger immune activation, and its properties outside the cell, remain poorly characterized. Three new preprints show roles for extracellular cGAMP and identify SLC19A1 as a transporter mediating uptake of extracellular cGAMP and other CDNs, allowing for immune activation.
Key findings:
- Luteijn et al and Cordova and Ritchie et al used loss-of-function CRISPR screens to identify SLC19A1 as the dominant cGAMP importer. The two preprints use distinct cell lines and readouts of CDN uptake to identify the cGAMP importer, but converge on SLC19A1. SLC19A1 is known to import folate, which – along with other known antagonists of SLC19A1 – is able to block uptake of CDNs and subsequent cellular activation. Bypassing the requirement for SLC19A1 by electroporation of CDNs or intracellular activation of cGAS resulted in normal activation in SLC19A1-deficient cells, confirming the role of SLC19A1 specifically in uptake of CDNs.
- Carozza et al developed a sensitive mass spectrometry approach to detect extracellular cGAMP. As they had previously shown that the extracellular enzyme ENPP1 (present in serum) cleaved cGAMP, they examined export by ENPP1-deficient cell lines in a serum-free culture. Cancer cell lines in an ENPP-1 free system constitutively exported cGAMP in a freely soluble form, at high levels. Inhibition of extracellular ENPP-1 with a novel cell-impermeable ENPP1 inhibitor increased extracellular cGAMP concentrations, with no effect on intracellular cGAMP. Finally, cGAMP secretion by cancer cells in vivo drove CD11c+ dendritic cell accumulation in the tumor, and inhibition of cGAMP degradation with the ENPP-1 inhibitor increased anti-tumor immunity.
Importance:
This trio of preprints greatly solidify our understanding of cGAMP activity and resolve important questions in the field regarding the activity of extracellular and exogenous cGAMP. This is particularly important as synthetic CDNs, and other mechanisms of perturbing the cGAS-STING pathway, move forward in clinical trials. Together, these papers help build a model of cGAMP export, extracellular stability, and import (Figure 1), providing multiple new avenues of study of CDN-mediated immunity and therapeutic axes to target this key immune pathway.
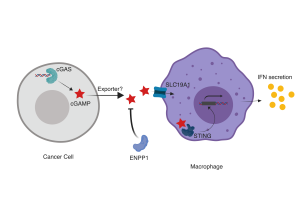
Figure 1: A model for extracellular cGAMP activity
Sensing of cytosolic dsDNA by cGAS leads to cGAMP synthesis in cancer cells. cGAMP is exported by an unknown mechanism, leading to free soluble extracellular cGAMP. This may be degraded by ENPP1, but can also be imported into immune cells such as macrophages by the transporter SLC19A1. There, it binds to STING, leading to IRF3 activation and downstream effects including type I IFN secretion.
Moving forward:
- The mechanism by which cGAMP becomes extracellular still remains unclear. Carozza et al suggest that cGAMP is efficiently exported from living cancer cells, rather than being released following cell death, but that suggests the existence of an exporter. The identification of this export mechanism, leading to freely soluble cGAMP (rather than transfer through gap junctions or membrane fusions, as has been previously demonstrated) will be important to improve our understanding of this pathway.
- In a similar vein, under what circumstances might CDN import be favorable in a non-tumor setting? It is demonstrated that SLC19A1 may also import bacterial CDNs, and Luteijn et al argue that pathogen- or microbiota-derived CDNs may play an important role in inflammatory diseases. Is cGAMP export common in viral infections, and might this be a pathway to avoiding viral STING antagonists (only operating in infected cells), or might bacterial CDNs be the “dominant” ligand driving the evolution of this pathway?
- Both Luteijn et al and Cordova and Ritchie et al show that SLC19A1 is the dominant cGAMP importer, however their data suggest the existence of other importers/ Luteijn et al examine other folate transporters, which show some ability to import CDNs but are still insufficient to explain the residual transport activity (although, the triple KD/KO of all three tested SLC transporters is not shown – perhaps that would lead to complete loss of CDN import?). Hopefully, further studies will identify the multiple pathways leading to CDN uptake.
Sign up to customise the site to your preferences and to receive alerts
Register hereAlso in the immunology category:
Gut microbiome changes over the course of multiple sclerosis differentially influence autoimmune neuroinflammation
Carole Djagang et al.
Human Assembloid Model of Emergent Neurotropic Enteroviruses
Mitchell Sarmie
Small Molecule Agonists of TREM2 Reprogram Microglia and Protect Synapses in Human Alzheimer’s Models
Dina Kabbara
preLists in the immunology category:
BSDB Spring Meeting: Molecules to Morphogenesis
The British Society for Developmental Biology (BSDB) Spring Meeting Molecules to Morphogenesis was held from 23–26 March 2026 at the University of Warwick (UK). This meeting brought together a vibrant community of researchers to discuss how molecular mechanisms are integrated across scales to drive morphogenesis, spanning diverse model systems and approaches. This preList contains preprints by presenters from the talk and poster sessions at the meeting. Please do get in touch at preLights@biologists.com if you notice any relevant preprints that we may have missed.
| List by | Ingrid Tsang |
SciELO preprints – From 2025 onwards
SciELO has become a cornerstone of open, multilingual scholarly communication across Latin America. Its preprint server, SciELO preprints, is expanding the global reach of preprinted research from the region (for more information, see our interview with Carolina Tanigushi). This preList brings together biological, English language SciELO preprints to help readers discover emerging work from the Global South. By highlighting these preprints in one place, we aim to support visibility, encourage early feedback, and showcase the vibrant research communities contributing to SciELO’s open science ecosystem.
| List by | Carolina Tanigushi |
Community-driven preList – Immunology
In this community-driven preList, a group of preLighters, with expertise in different areas of immunology have worked together to create this preprint reading list.
| List by | Felipe Del Valle Batalla et al. |
Journal of Cell Science meeting ‘Imaging Cell Dynamics’
This preList highlights the preprints discussed at the JCS meeting 'Imaging Cell Dynamics'. The meeting was held from 14 - 17 May 2023 in Lisbon, Portugal and was organised by Erika Holzbaur, Jennifer Lippincott-Schwartz, Rob Parton and Michael Way.
| List by | Helen Zenner |
Fibroblasts
The advances in fibroblast biology preList explores the recent discoveries and preprints of the fibroblast world. Get ready to immerse yourself with this list created for fibroblasts aficionados and lovers, and beyond. Here, my goal is to include preprints of fibroblast biology, heterogeneity, fate, extracellular matrix, behavior, topography, single-cell atlases, spatial transcriptomics, and their matrix!
| List by | Osvaldo Contreras |
Single Cell Biology 2020
A list of preprints mentioned at the Wellcome Genome Campus Single Cell Biology 2020 meeting.
| List by | Alex Eve |
Autophagy
Preprints on autophagy and lysosomal degradation and its role in neurodegeneration and disease. Includes molecular mechanisms, upstream signalling and regulation as well as studies on pharmaceutical interventions to upregulate the process.
| List by | Sandra Malmgren Hill |
Antimicrobials: Discovery, clinical use, and development of resistance
Preprints that describe the discovery of new antimicrobials and any improvements made regarding their clinical use. Includes preprints that detail the factors affecting antimicrobial selection and the development of antimicrobial resistance.
| List by | Zhang-He Goh |
Zebrafish immunology
A compilation of cutting-edge research that uses the zebrafish as a model system to elucidate novel immunological mechanisms in health and disease.
| List by | Shikha Nayar |











 (No Ratings Yet)
(No Ratings Yet)