No global collapse of food webs across the Permian–Triassic Mass Extinction
Posted on: 3 May 2026
Preprint posted on 25 February 2026
Ancient webs of life; The authors of the study evaluate the effect of the Permian-Triassic mass extinction (PTME) on marine food-webs and post-extinction recovery.
Selected by Theodora StougiannouCategories: ecology, evolutionary biology, paleontology
Ancient webs of life; The authors of the study evaluate the effect of the Permian-Triassic mass extinction (PTME) on marine food-webs and post-extinction recovery.
Life on Earth: Mass extinctions
A mass extinction event can be generally defined as an event during which a large percentage of higher taxa in many different biological groups disappears; this disappearance generally occurs within a relatively small timeframe, on a global scale. However, no universally accepted quantitative definition for mass extinctions yet exists. Often referred to as ‘pulses of extinction’, these often occur near or at the ends of formally recognized geological periods.
Regarding marine extinctions, the ‘pulses of extinction model is the one commonly accepted. Some, however, posit that there is indeed evidence for a large percentage of extinctions occurring as pulses towards the end of geological periods, following a period of significant amount of background extinction [1] (Stanley et al., 2016).
Mass extinctions result in loss of species and can be followed by rapid speciation/increased diversity called adaptive radiation; this results in changes in the fossil record, with mass extinctions often defining boundaries between geological periods in the story of life on Earth.
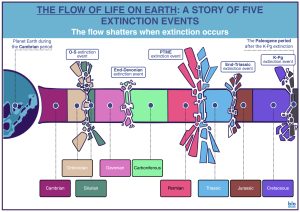
The Permian-Triassic mass extinction (PTME)
PTME, or as alternatively known, P/T, is a mass extinction event that occurred at the end of the Permian period. While some theorize the event occurred as a series of extinction pulses across a timeframe of 15 million years during the late Permian period, others maintain that the event occurred across a much smaller timeframe, of about 200,000 years.
Based on fossil data found in South China, it is believed that the effects of PTME on marine food webs included truncation of the upper trophic levels, followed by recovery proceeding gradually from primary producers upwards. Despite this generalization, however, groups of aquatic animal groups capable of swimming independent of water currents (nektonic) present in upper levels of the food chains (fishes, ammonoids) are less affected compared to animals groups with limited or no-mobility (non-motile) usually inhabiting the sea floor (benthic groups such as foraminifers, brachiopods) [2] (Karapunar et al., 2026) [3] (Jurikova et al., 2020).
Some questions thus remain:
- Was the disruption to these trophic systems (food chains) global, or did it vary based on location?
- Did the marine food webs collapse from the top levels (top-down collapse)? Did recovery after the PTME (P/T) occur from the bottom-up?
- Did the marine food webs reorganize in the same manner across different locations, or was the reorganization after the extinction also varied depending on geographical location? a uniform, or spatially variable manner?
What are the preprint authors looking at?
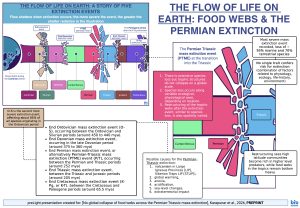
The study by Karapunar and team seeks to characterize the loss occurring in marine food webs and the subsequently recovery, following the PTME (or P/T) extinction event. To this end they recreated food webs based on fossil trait data and simulated 17 extinction scenarios; based on these, both direct species losses, as well as secondary losses due to cascading effects could be estimated.
Key aspects of the study
- Though there is extensive species loss across all the sites studied, overall, the trophic structures do not globally collapse; as such, most areas retained 4 trophic levels throughout the extinction event. Trophic levels denote hierarchical steps, that all together constitute a food chain; primary producers, i.e., organisms that generate their own nutrition using either sunlight (photosynthesis), or chemical energy (chemosynthesis) usually constitute the first trophic level. The second level will usually be taken up by primary consumers, organisms that feed exclusively on these primary producers.
- Even as species diversity was lost, higher trophic levels were maintained in almost all the sites evaluated.
- Upper trophic levels were still present in food web communities originating in mid- and high latitudes; loss of marine predators was not global.
- Based on extinction scenario simulations, it could be seen that species loss occurred along variable ecological and physiological axes, depending on location. This shows that different physiological traits were advantageous for survival in different environment during the extinction event, and advantageous versus disadvantageous traits are uniform globally. Previous studies suggested that a single global axis exists, along which species were lost, comprising traits such as low physiological buffering capacity, limiter motility, low respiratory capacity, large size.
- For example, in high-latitude locations (Greenland, Tibet), limited motility was the axis along which species were lost, with preferential loss of non-motile organisms mostly living on the sea floor (benthic).
- In samples from Meishan and the Kashmir, it was observed that loss occurred along the physiological trait of respiratory capacity, with species characterized by low respiratory capacity (foraminifera, echinoderms, bryozoans) affected the most. Extinctions occurring in the Kashmir, however, also include a strong random component as well.
- Species loss along this physiological trait axis (respiratory capacity) also occurred in the tropical Dolomites; in this case, however, taxa with high respiratory capacity were lost instead, such as vertebrates, cephalopods [2] (Karapunar et al., 2026).
- Despite regional variability, some trends in primary extinction selectivity could be observed; namely, it affected primarily taxa with limited motility and physiological constraints, especially non-motile benthic organisms that function as primary consumers and are further characterized by low respiratory capacity (Foraminifera, Brachiopoda, Bryozoa, Cnidaria).
- Species occupying the higher trophic levels, such as nektonic predators found in middle to higher latitude communities (fishes, conodonts) were relatively less affected; this could also explain the rapid diversification occurring in these species post-extinction.
- No single trait confers risk for extinction, as this is mediated by a combination of factors relating to physiology, ecology, life-history and local environmental factors [2] (Karapunar et al., 2026).
- Restructuring of the trophic webs after the extinction event, similar to species loss, was also spatially varied.
- For example, samples from areas in low latitudes (Meishan, Türkiye, Dolomites), exhibit mainly retention of bottom trophic levels, despite losses occurring in benthic primary consumers. These tropic food webs were also more vulnerable to secondary extinction cascades.
- Samples from areas found in mid- to higher latitudes (Tiber, Kashmir, Greenland), show that communities in these locations became increasingly top-heavy, as higher trophic levels of the food webs dominated in these areas.
- This could be explained by both newly evolved species as well as species migrating from other locations. In fact, in these higher latitude systems, entry of predatory fishes, temnospondyls, and cephalopods produced quaternary and quinary trophic levels, increasing both mean and maximum trophic levels in the process.
Why this work is interesting
The work by Karapunar and team offers a glimpse into one of the most severe mass extinction events on this planet; it also shows how the trophic networks reorganize and recover. Additionally, it reflects on how spatially varied restructuring in the trophic web can possibly be affected by distinct elements in the local environments, while at the same time, it helps scientists anticipate how marine ecosystems might behave in cases of modern environmental changes.
For me, this preprint offers a glimpse into different locations across the planet at a time when a severe mass extinction was taking place, severe enough to change the geological record and lead to a new era, the Triassic. How were the marine ecosystems of the time responding to this devastation, across the world? An infographic summary of the main information of the article, along with interesting background information follows.
Glossary

Questions to the authors
- In your study, you utilize samples across 7 regions to evaluate the nature of the effects triggered by the PTME, as well as the response and restructuring of the communities following the event. How many samples do you think is ‘enough’ to have an accurate idea about the state of marine food webs at that particular time during the life history of Earth?
- The fact that primary extinction pressures are varied depending on geographical location is associated with extinction selectivity across a wide variety of physiological traits. Do you think that this variety in primary extinction pressures across different locations reflects the different changing factors in the local environment that was shaping the planet at the time, and in turn, led to this mass extinction event?
References
[1] Stanley SM. Estimates of the magnitudes of major marine mass extinctions in earth history. Proceedings of the National Academy of Sciences 2016;113:E6325–34. https://doi.org/10.1073/pnas.1613094113.
[2 Karapunar B, Strydom T, Beckerman AP, Ridgwell A, Wignall PB, Dunne JA, et al. No global collapse of food webs across the Permian–Triassic Mass Extinction 2026:2026.02.24.707709. https://doi.org/10.64898/2026.02.24.707709.
[3] Jurikova H, Gutjahr M, Wallmann K, Flögel S, Liebetrau V, Posenato R, et al. Permian–Triassic mass extinction pulses driven by major marine carbon cycle perturbations. Nat Geosci 2020;13:745–50. https://doi.org/10.1038/s41561-020-00646-4.
doi: https://doi.org/10.1242/prelights.43367
Read preprintSign up to customise the site to your preferences and to receive alerts
Register hereAlso in the ecology category:
Heatwave winners and losers: cryptic coral holobionts differ in thermal tolerance
Maitri Manjunath
Resilience to cardiac aging in Greenland shark Somniosus microcephalus
Theodora Stougiannou
Cannibalism as a mechanism to offset reproductive costs in three-spined sticklebacks
Tina Nguyen
Also in the evolutionary biology category:
Heatwave winners and losers: cryptic coral holobionts differ in thermal tolerance
Maitri Manjunath
No global collapse of food webs across the Permian–Triassic Mass Extinction
Theodora Stougiannou
Remote homology and functional genetics unmask deeply preserved Scm3/HJURP orthologs in metazoans
Reinier Prosee
Also in the paleontology category:
No global collapse of food webs across the Permian–Triassic Mass Extinction
Theodora Stougiannou
A high-coverage genome from a 200,000-year-old Denisovan
AND
A global map for introgressed structural variation and selection in humans
Siddharth Singh
Evaluating the impact and detectability of mass extinctions on total-evidence dating
Tom Carruthers
preLists in the ecology category:
SciELO preprints – From 2025 onwards
SciELO has become a cornerstone of open, multilingual scholarly communication across Latin America. Its preprint server, SciELO preprints, is expanding the global reach of preprinted research from the region (for more information, see our interview with Carolina Tanigushi). This preList brings together biological, English language SciELO preprints to help readers discover emerging work from the Global South. By highlighting these preprints in one place, we aim to support visibility, encourage early feedback, and showcase the vibrant research communities contributing to SciELO’s open science ecosystem.
| List by | Carolina Tanigushi |
November in preprints – DevBio & Stem cell biology
preLighters with expertise across developmental and stem cell biology have nominated a few developmental and stem cell biology (and related) preprints posted in November they’re excited about and explain in a single paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Aline Grata et al. |
Biologists @ 100 conference preList
This preList aims to capture all preprints being discussed at the Biologists @100 conference in Liverpool, UK, either as part of the poster sessions or the (flash/short/full-length) talks.
| List by | Reinier Prosee, Jonathan Townson |
preLights peer support – preprints of interest
This is a preprint repository to organise the preprints and preLights covered through the 'preLights peer support' initiative.
| List by | preLights peer support |
EMBO | EMBL Symposium: The organism and its environment
This preList contains preprints discussed during the 'EMBO | EMBL Symposium: The organism and its environment', organised at EMBL Heidelberg, Germany (May 2023).
| List by | Girish Kale |
Bats
A list of preprints dealing with the ecology, evolution and behavior of bats
| List by | Baheerathan Murugavel |
Also in the evolutionary biology category:
SciELO preprints – From 2025 onwards
SciELO has become a cornerstone of open, multilingual scholarly communication across Latin America. Its preprint server, SciELO preprints, is expanding the global reach of preprinted research from the region (for more information, see our interview with Carolina Tanigushi). This preList brings together biological, English language SciELO preprints to help readers discover emerging work from the Global South. By highlighting these preprints in one place, we aim to support visibility, encourage early feedback, and showcase the vibrant research communities contributing to SciELO’s open science ecosystem.
| List by | Carolina Tanigushi |
November in preprints – DevBio & Stem cell biology
preLighters with expertise across developmental and stem cell biology have nominated a few developmental and stem cell biology (and related) preprints posted in November they’re excited about and explain in a single paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Aline Grata et al. |
October in preprints – DevBio & Stem cell biology
Each month, preLighters with expertise across developmental and stem cell biology nominate a few recent developmental and stem cell biology (and related) preprints they’re excited about and explain in a single paragraph why. Short, snappy picks from working scientists — a quick way to spot fresh ideas, bold methods and papers worth reading in full. These preprints can all be found in the October preprint list published on the Node.
| List by | Deevitha Balasubramanian et al. |
October in preprints – Cell biology edition
Different preLighters, with expertise across cell biology, have worked together to create this preprint reading list for researchers with an interest in cell biology. This month, most picks fall under (1) Cell organelles and organisation, followed by (2) Mechanosignaling and mechanotransduction, (3) Cell cycle and division and (4) Cell migration
| List by | Matthew Davies et al. |
Biologists @ 100 conference preList
This preList aims to capture all preprints being discussed at the Biologists @100 conference in Liverpool, UK, either as part of the poster sessions or the (flash/short/full-length) talks.
| List by | Reinier Prosee, Jonathan Townson |
‘In preprints’ from Development 2022-2023
A list of the preprints featured in Development's 'In preprints' articles between 2022-2023
| List by | Alex Eve, Katherine Brown |
preLights peer support – preprints of interest
This is a preprint repository to organise the preprints and preLights covered through the 'preLights peer support' initiative.
| List by | preLights peer support |
EMBO | EMBL Symposium: The organism and its environment
This preList contains preprints discussed during the 'EMBO | EMBL Symposium: The organism and its environment', organised at EMBL Heidelberg, Germany (May 2023).
| List by | Girish Kale |
9th International Symposium on the Biology of Vertebrate Sex Determination
This preList contains preprints discussed during the 9th International Symposium on the Biology of Vertebrate Sex Determination. This conference was held in Kona, Hawaii from April 17th to 21st 2023.
| List by | Martin Estermann |
EMBL Synthetic Morphogenesis: From Gene Circuits to Tissue Architecture (2021)
A list of preprints mentioned at the #EESmorphoG virtual meeting in 2021.
| List by | Alex Eve |
Planar Cell Polarity – PCP
This preList contains preprints about the latest findings on Planar Cell Polarity (PCP) in various model organisms at the molecular, cellular and tissue levels.
| List by | Ana Dorrego-Rivas |
TAGC 2020
Preprints recently presented at the virtual Allied Genetics Conference, April 22-26, 2020. #TAGC20
| List by | Maiko Kitaoka et al. |
ECFG15 – Fungal biology
Preprints presented at 15th European Conference on Fungal Genetics 17-20 February 2020 Rome
| List by | Hiral Shah |
COVID-19 / SARS-CoV-2 preprints
List of important preprints dealing with the ongoing coronavirus outbreak. See http://covidpreprints.com for additional resources and timeline, and https://connect.biorxiv.org/relate/content/181 for full list of bioRxiv and medRxiv preprints on this topic
| List by | Dey Lab, Zhang-He Goh |
1
SDB 78th Annual Meeting 2019
A curation of the preprints presented at the SDB meeting in Boston, July 26-30 2019. The preList will be updated throughout the duration of the meeting.
| List by | Alex Eve |
Pattern formation during development
The aim of this preList is to integrate results about the mechanisms that govern patterning during development, from genes implicated in the processes to theoritical models of pattern formation in nature.
| List by | Alexa Sadier |











 (No Ratings Yet)
(No Ratings Yet)