Immune factor of bacterial origin protects ticks against host microbial commensals
Posted on: 13 May 2020
Preprint posted on 11 April 2020
Article now published in Cell at http://dx.doi.org/10.1016/j.cell.2020.10.042
How Tick Spit got its Bite – Tick Immunity Using a Bacterially-Derived Protein
Selected by Connor RosenCategories: evolutionary biology, microbiology
Background:
Horizontal gene transfer is a dominant mode of genome evolution in bacteria, but examples of cross-kingdom transfer of genes from bacteria into animals are rare. Previous work identified a family of anti-bacterial effectors likely transferred from bacteria to a common ancestor of ticks and mites hundreds of millions of years ago [Chou and Daugherty et al, 2014]. The adaptation of bacterial Type 6 secretion amidase effectors (TAEs) into domesticated amidase effectors (DAEs) suggested they may serve a beneficial effect in the new host organism. The tick Dae2 maintains anti-microbial activity, but its role in tick fitness remained unclear. In this preprint, Hayes and Radkov et al illuminate in more detail the biochemical activity of Dae2, and identify a role for its activity during tick bloodfeeding.
Key findings:
- A horizontally-transferred amidase effector in ticks has expanded peptidoglycan specificity and direct antibacterial activity
Comparing Ixodes scapularis Dae2 to a bacterial homolog (Tae2 from Salmonella enterica) revealed that while Tae2 shows specificity towards Gram-negative cell wall peptidoglycan, Dae2 has an expanded substrate range and can degrade Gram-positive cell wall peptidoglycans as well. This corresponds to an increased anti-microbial activity, as Dae2 can directly kill several Gram-positive bacteria in culture, including multiple human skin commensal species.
- Dae2 enhances tick fitness during feeding and infection
The authors found that Dae2 was upregulated during bloodfeeding and injected into the host in the tick saliva. This suggested it could target commensal skin microbes in the host, which might represent novel pathogens that could infect the tick during feeding. Blocking Dae2 with gene silencing or pre-bite passive immunization of the mouse host increased the colonization of Staphylococcus species in the ticks, as well as decreased the feeding efficiency of the tick. Additional experiments suggested that increased Staphylococcus colonization of ticks was pathogenic, leading to tick death when Dae2 activity was blocked. This activity is summarized in Figure 1, below.
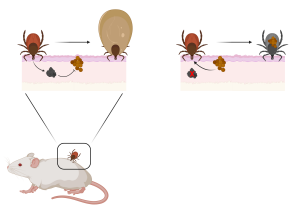
Figure 1: The role of horizontally-acquired Dae2 during tick bloodfeeding.
During tick bloodfeeding, the Dae2 protein is injected into the host where it can target and kill host commensal skin microbes, such as Staphylococcus species. This enables robust feeding by the tick. In the absence of Dae2 activity (right), host commensal species can colonize the tick gut and decrease tick feeding and fitness. Figure prepared with Biorender.
Importance:
This study offers a compelling case study explaining a rare example of bacteria-animal horizontal gene transfer. The clear fitness benefit identified of Dae2 in ticks shows how acquisition of bacterial genes could be selected for over long evolutionary time and the potential of bacterial genes to expand the immune capabilities of animal hosts. Additionally, this study identifies a new biological pressure at the host-vector-microbe interface, where host commensals may act as vector pathogens – analogous to how vectors may harbor numerous host pathogens. Overall, this study expands both our understanding of how horizontal gene transfer may be used during animal evolution, as well as opens a new avenue to studying host-vector biology and the complex role of host microbes in shaping this interaction.
Moving Forward / Questions for Authors:
- Dae2 is present in other tick species as well. It would be interesting to see how Dae2 antimicrobial activity has expanded or changed dependent on the host range of those ticks, and therefore the commensal microbes they are exposed to.
- Ticks and other hematophagous vectors inject many proteins during bloodfeeding. Given that Dae2 has direct activity only against Gram-positive microbes, since it has no ability to cross the outer membrane of Gram-negatives, are there other effectors it might “cooperate” with to target more microbes? Do ticks have a broader range of anti-microbial salivary proteins than previously anticipated? The scope of tick (and vector) saliva targeting of host commensals, rather than the host directly, will be interesting to see, as the host-vector-microbe triangle expands.
References:
- Chou S and Daugherty MD et al, “Transferred interbacterial antagonism genes augment eukaryotic innate immune function”, Nature (2014) 518:98-101
doi: https://doi.org/10.1242/prelights.20604
Read preprintSign up to customise the site to your preferences and to receive alerts
Register hereAlso in the evolutionary biology category:
Heatwave winners and losers: cryptic coral holobionts differ in thermal tolerance
Maitri Manjunath
No global collapse of food webs across the Permian–Triassic Mass Extinction
Theodora Stougiannou
Remote homology and functional genetics unmask deeply preserved Scm3/HJURP orthologs in metazoans
Reinier Prosee
Also in the microbiology category:
Gut microbiome changes over the course of multiple sclerosis differentially influence autoimmune neuroinflammation
Carole Djagang et al.
Human Assembloid Model of Emergent Neurotropic Enteroviruses
Mitchell Sarmie
Cryo-EM reveals multiple mechanisms of ribosome inhibition by doxycycline
Leonie Brüne
preLists in the evolutionary biology category:
SciELO preprints – From 2025 onwards
SciELO has become a cornerstone of open, multilingual scholarly communication across Latin America. Its preprint server, SciELO preprints, is expanding the global reach of preprinted research from the region (for more information, see our interview with Carolina Tanigushi). This preList brings together biological, English language SciELO preprints to help readers discover emerging work from the Global South. By highlighting these preprints in one place, we aim to support visibility, encourage early feedback, and showcase the vibrant research communities contributing to SciELO’s open science ecosystem.
| List by | Carolina Tanigushi |
November in preprints – DevBio & Stem cell biology
preLighters with expertise across developmental and stem cell biology have nominated a few developmental and stem cell biology (and related) preprints posted in November they’re excited about and explain in a single paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Aline Grata et al. |
October in preprints – DevBio & Stem cell biology
Each month, preLighters with expertise across developmental and stem cell biology nominate a few recent developmental and stem cell biology (and related) preprints they’re excited about and explain in a single paragraph why. Short, snappy picks from working scientists — a quick way to spot fresh ideas, bold methods and papers worth reading in full. These preprints can all be found in the October preprint list published on the Node.
| List by | Deevitha Balasubramanian et al. |
October in preprints – Cell biology edition
Different preLighters, with expertise across cell biology, have worked together to create this preprint reading list for researchers with an interest in cell biology. This month, most picks fall under (1) Cell organelles and organisation, followed by (2) Mechanosignaling and mechanotransduction, (3) Cell cycle and division and (4) Cell migration
| List by | Matthew Davies et al. |
Biologists @ 100 conference preList
This preList aims to capture all preprints being discussed at the Biologists @100 conference in Liverpool, UK, either as part of the poster sessions or the (flash/short/full-length) talks.
| List by | Reinier Prosee, Jonathan Townson |
‘In preprints’ from Development 2022-2023
A list of the preprints featured in Development's 'In preprints' articles between 2022-2023
| List by | Alex Eve, Katherine Brown |
preLights peer support – preprints of interest
This is a preprint repository to organise the preprints and preLights covered through the 'preLights peer support' initiative.
| List by | preLights peer support |
EMBO | EMBL Symposium: The organism and its environment
This preList contains preprints discussed during the 'EMBO | EMBL Symposium: The organism and its environment', organised at EMBL Heidelberg, Germany (May 2023).
| List by | Girish Kale |
9th International Symposium on the Biology of Vertebrate Sex Determination
This preList contains preprints discussed during the 9th International Symposium on the Biology of Vertebrate Sex Determination. This conference was held in Kona, Hawaii from April 17th to 21st 2023.
| List by | Martin Estermann |
EMBL Synthetic Morphogenesis: From Gene Circuits to Tissue Architecture (2021)
A list of preprints mentioned at the #EESmorphoG virtual meeting in 2021.
| List by | Alex Eve |
Planar Cell Polarity – PCP
This preList contains preprints about the latest findings on Planar Cell Polarity (PCP) in various model organisms at the molecular, cellular and tissue levels.
| List by | Ana Dorrego-Rivas |
TAGC 2020
Preprints recently presented at the virtual Allied Genetics Conference, April 22-26, 2020. #TAGC20
| List by | Maiko Kitaoka et al. |
ECFG15 – Fungal biology
Preprints presented at 15th European Conference on Fungal Genetics 17-20 February 2020 Rome
| List by | Hiral Shah |
COVID-19 / SARS-CoV-2 preprints
List of important preprints dealing with the ongoing coronavirus outbreak. See http://covidpreprints.com for additional resources and timeline, and https://connect.biorxiv.org/relate/content/181 for full list of bioRxiv and medRxiv preprints on this topic
| List by | Dey Lab, Zhang-He Goh |
1
SDB 78th Annual Meeting 2019
A curation of the preprints presented at the SDB meeting in Boston, July 26-30 2019. The preList will be updated throughout the duration of the meeting.
| List by | Alex Eve |
Pattern formation during development
The aim of this preList is to integrate results about the mechanisms that govern patterning during development, from genes implicated in the processes to theoritical models of pattern formation in nature.
| List by | Alexa Sadier |
Also in the microbiology category:
SciELO preprints – From 2025 onwards
SciELO has become a cornerstone of open, multilingual scholarly communication across Latin America. Its preprint server, SciELO preprints, is expanding the global reach of preprinted research from the region (for more information, see our interview with Carolina Tanigushi). This preList brings together biological, English language SciELO preprints to help readers discover emerging work from the Global South. By highlighting these preprints in one place, we aim to support visibility, encourage early feedback, and showcase the vibrant research communities contributing to SciELO’s open science ecosystem.
| List by | Carolina Tanigushi |
BioMalPar XVI: Biology and Pathology of the Malaria Parasite
[under construction] Preprints presented at the (fully virtual) EMBL BioMalPar XVI, 17-18 May 2020 #emblmalaria
| List by | Dey Lab, Samantha Seah |
1
ECFG15 – Fungal biology
Preprints presented at 15th European Conference on Fungal Genetics 17-20 February 2020 Rome
| List by | Hiral Shah |
EMBL Seeing is Believing – Imaging the Molecular Processes of Life
Preprints discussed at the 2019 edition of Seeing is Believing, at EMBL Heidelberg from the 9th-12th October 2019
| List by | Dey Lab |
Antimicrobials: Discovery, clinical use, and development of resistance
Preprints that describe the discovery of new antimicrobials and any improvements made regarding their clinical use. Includes preprints that detail the factors affecting antimicrobial selection and the development of antimicrobial resistance.
| List by | Zhang-He Goh |











 (No Ratings Yet)
(No Ratings Yet)