Atropselective Oxidation of 2,2',3,3',4,6'-Hexachlorobiphenyl (PCB 132) to Hydroxylated Metabolites by Human Liver Microsomes: Involvement of Arene Oxide Intermediates
Posted on: 13 August 2018 , updated on: 29 September 2019
Preprint posted on 23 July 2018
Article now published in Toxicological Sciences at http://dx.doi.org/10.1093/toxsci/kfz150
Through the looking glass: Characterising the atropselective oxidation of PCBs by Human Liver Microsomes
Selected by Zhang-He GohCategories: pharmacology and toxicology
Background of preprint
Compounds in the polychlorinated biphenyl (PCB) family have often been implicated in various toxicities, especially neurological disorders. This is worrying for three reasons. First, mounting toxicological evidence points to an association between PCBs and behavioural disturbances in both in vivo rodent studies as well as etiological studies. Second, the ubiquitous nature of PCBs, which stems from their extensive use in manufacturing prior to their 1979 US ban, means that human and environmental exposure to them is inevitable. Third, despite the threat that PCBs pose to human and environmental health, relatively little is understood about how they are metabolised in the body.
Like pharmacological studies, toxicological studies are built on two main pillars: Toxicodynamics, the study of how ligand-target interactions lead to toxicity; and toxicokinetics, the study of how the body’s handling of the toxicant translates to noxious outcomes. The toxicodynamics of the PCB family are relatively well understood, but the toxicokinetics of the PCB family and its implications on toxicity are not as widely researched. In this study, Uwimana et al aimed to bridge this research gap by studying the metabolism of PCB 132 and its structurally-related congeners, which have been detected in both the air and in food sources. Specifically, Uwimana et al investigated the metabolism of PCB 132 to its hydroxylated metabolites (OH-PCB) by Human Liver Microsomes (HLM).
Key findings of preprint
Uwimana et al focused on the investigation of the major metabolites 3’-140 and 5’-132 (Figure 1) in their study on the metabolism of PCB 132.
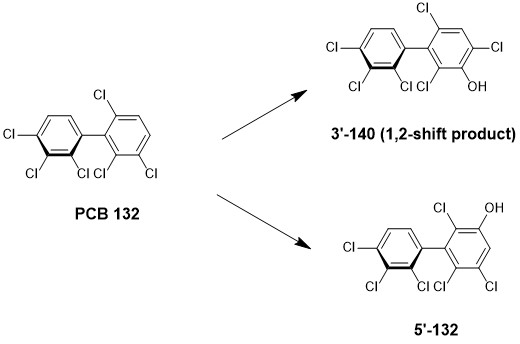
Figure 1. Metabolism of PCB 132 in incubation with HLM
This study corroborates findings of previous studies in two aspects. First, it illustrates that the metabolism of PCB 132 to the 1,2-shift product 3’-140 occurs via an arene oxide intermediate. Second, the position at which PCB 132 is metabolised also supports the current paradigm of structure-metabolism relationships established for the oxidation of PCB 132. At the same time, the findings from this study also contrast with studies demonstrating that 5’-132 is a major metabolite of PCB 132 in rodents, a juxtaposition which underscores the importance of recognising interspecies differences in biological research.
Mechanistic studies of energetics underlying the atropselectivity of the metabolism by reducing the PCBs to the simplest case—1,2,4-trichlorobenzene—also show that the metabolism of PCB relies mainly on the energetics involving just the 2,3,6-trichlorobenzene ring. At the same time, these explain the regioselective formation of only the 3’ and 6’ products over the 4’ and 5’ products as well.
This study of the metabolism of PCB 132 using HLMs also has three clinical implications:
- The metabolism of PCB 132 exhibits interindividual variation;
- The enrichment of PCB atropisomers observed in vitro may predict the atropisomeric enrichment observed in animal studies, suggesting that the atropisomeric enrichment seen in humans is caused by the selective metabolism of certain atropisomers over others; and
- The selectivity in metabolising certain atropisomers over others also leads to the atropselective formation of particular hydroxylated metabolites at varying rates.
What I like about this preprint
I chose this preprint for two reasons. The first is that it answers the intriguing question of how PCBs are metabolised. While this question has traditionally been answered using animal models, interspecies differences in metabolism make it challenging for researchers to apply the conclusions in animal work to humans. By using HLM in this work to profile the metabolites of PCB 132 in vitro, Uwimana et al sidestep this difficulty. Furthermore, such an investigation also yields the opportunity to investigate the causes behind the differences in atropselectivity for these metabolic reactions.
The second reason for choosing this preprint is that it complements two articles by Uwimana et al earlier this year about PCB 91 [1] and PCB 95 [2], allowing Uwimana et al to compare across these members of the PCB family. Like PCB 132, the metabolism of PCB 91 also involves the formation of an arene oxide intermediate followed by a 1,2-chloride shift, whose energetics are largely determined by just the 2,3,6-trichlorobenzene ring. In contrast, PCB 95 was preferentially oxidised in the para position, and the 1,2-shift product was formed as the minor product.
Future directions
This study adds to the previous work published by Uwimana et al and other researchers in understanding the nature of various PCBs. Comparing between the members of the PCB family aids in our understanding of how minute structural changes can lead to significant differences in the toxicokinetics of these compounds. At the same time, the progress in our current knowledge on the toxicodynamics of PCBs will also paint a clearer picture on the extent of the threat that PCBs pose to human and environmental health.
Future work in this field will likely involve comparing the toxicokinetics between the PCBs and expanding our knowledge of their atropselective metabolism with regards to the metabolising enzymes. These studies will be useful in two ways. The first route is more direct—a better understanding of the toxicokinetics of different PCBs will help industry players to reduce risks in using certain members of the PCB family. The atropselectivity in PCB metabolism of members suggests that the more toxic compounds may be replaced with safer alternatives, thus enhancing product safety to employees as well as consumers.
The other benefit of such a study is more indirect. The close relationship between pharmacology and toxicology allows researchers to easily apply the knowledge gained in one field to the other. Therefore, a more thorough understanding of metabolising enzymes will also enable pharmacologists to design drugs that exhibit improved pharmacokinetics, leading to better safety and efficacy.
Questions for the authors
The reasons for the formation of the para substituted metabolites of PCB 95 and PCB 136 extend beyond the energetics of the arene oxide intermediate to include steric and electronic interactions. Is it possible that, in the case of PCB 95 and PCB 136, the chirality of the PCB plays a role here? Given that the 1,2-chloride shift products of PCB 132 and PCB 91 are not influenced by the second aromatic ring, was the observation that PCB 95 and PCB 136 would demonstrate such a different property expected?
References
[1] Uwimana E, Li X, Lehmler H-J, Human Liver Microsomes Atropselectively Metabolize 2,2′,3,4′,6-Pentachlorobiphenyl (PCB 91) to a 1,2-Shift Product as the Major Metabolite, Environmental Science & Technology 52(10) (2018) 6000-6008.
[2] Uwimana E, Li X, Lehmler H-J, 2,2′,3,5′,6-Pentachlorobiphenyl (PCB 95) Is Atropselectively Metabolized to para-Hydroxylated Metabolites by Human Liver Microsomes, Chemical Research in Toxicology 29(12) (2016) 2108-2110.
doi: https://doi.org/10.1242/prelights.4370
Read preprintSign up to customise the site to your preferences and to receive alerts
Register hereAlso in the pharmacology and toxicology category:
Small Molecule Agonists of TREM2 Reprogram Microglia and Protect Synapses in Human Alzheimer’s Models
Dina Kabbara
Snake venom metalloproteinases are predominantly responsible for the cytotoxic effects of certain African viper venoms
Daniel Osorno Valencia
Cryo-EM reveals multiple mechanisms of ribosome inhibition by doxycycline
Leonie Brüne
preLists in the pharmacology and toxicology category:
SciELO preprints – From 2025 onwards
SciELO has become a cornerstone of open, multilingual scholarly communication across Latin America. Its preprint server, SciELO preprints, is expanding the global reach of preprinted research from the region (for more information, see our interview with Carolina Tanigushi). This preList brings together biological, English language SciELO preprints to help readers discover emerging work from the Global South. By highlighting these preprints in one place, we aim to support visibility, encourage early feedback, and showcase the vibrant research communities contributing to SciELO’s open science ecosystem.
| List by | Carolina Tanigushi |
April in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) biochemistry/metabolism 2) cell cycle and division 3) cell organelles and organisation 4) cell signalling and mechanosensing 5) (epi)genetics
| List by | Vibha SINGH et al. |
FENS 2020
A collection of preprints presented during the virtual meeting of the Federation of European Neuroscience Societies (FENS) in 2020
| List by | Ana Dorrego-Rivas |
COVID-19 / SARS-CoV-2 preprints
List of important preprints dealing with the ongoing coronavirus outbreak. See http://covidpreprints.com for additional resources and timeline, and https://connect.biorxiv.org/relate/content/181 for full list of bioRxiv and medRxiv preprints on this topic
| List by | Dey Lab, Zhang-He Goh |
1
Drug use in special populations
Any drugs that are being used in special populations: Patients with liver and kidney failure, in paediatrics, in geriatrics, and in pregnant or lactating patients. Includes the discovery of factors that could potentially affect drug use in these special populations.
| List by | Zhang-He Goh |
Toxicology of toxicants, existing therapeutics, and investigational drugs
Preprints that describe the toxicology of environmental pollutants and existing and upcoming drugs. Includes both toxicokinetics and toxicodynamics, as well as technological improvements that will help in the characterisation of this field.
| List by | Zhang-He Goh |
Antimicrobials: Discovery, clinical use, and development of resistance
Preprints that describe the discovery of new antimicrobials and any improvements made regarding their clinical use. Includes preprints that detail the factors affecting antimicrobial selection and the development of antimicrobial resistance.
| List by | Zhang-He Goh |
Anticancer agents: Discovery and clinical use
Preprints that describe the discovery of anticancer agents and their clinical use. Includes both small molecules and macromolecules like biologics.
| List by | Zhang-He Goh |
Advances in Drug Delivery
Advances in formulation technology or targeted delivery methods that describe or develop the distribution of small molecules or large macromolecules to specific parts of the body.
| List by | Zhang-He Goh |











 (1 votes)
(1 votes)