Mouse dLGN receives input from a diverse population of retinal ganglion cells with limited convergence
Posted on: 4 July 2018
Preprint posted on 15 May 2018
Article now published in Neuron at http://dx.doi.org/10.1016/j.neuron.2019.01.040
How much does the thalamus process visual information before it reaches the cortex? This preprint from Román Rosón et al. sheds light on this question by looking at the functional connectivity between the retina and the thalamus.
Selected by Polona JagerCategories: neuroscience
Background
In vertebrates, visual perception begins in the retina of the eye, and this information is relayed to the brain by retinal output neurons – the retinal ganglion cells (RGCs). There exist at least 30 distinct RGC types, each transmitting information about a specific visual feature.
In the mammalian brain RGCs transfer information to more than 50 different regions, including the dorsal lateral geniculate nucleus (dLGN) in the thalamus. The dLGN extensively connects with the primary visual cortex, and is part of the pathway that gives rise to conscious visual perception or sight.
How then are visual inputs from the retina processed in the thalamus, the first stop in the brain’s visual pathway? To answer this question, studies have looked at “RGC convergence” – the number and diversity of RGCs that project to individual dLGN thalamocortical (TC) neurons. Low convergence would suggest a more relay-like transmission, whereas high convergence implies that a more complex transformation or integration of visual information may take place in the thalamus, before reaching the cortex.
Prior physiological work in the dLGN across mammalian species suggested only limited RGC convergence onto target dLGN cells. However, more recent structural electron microscopy imaging, transsynaptic tracing, and functional calcium imaging of retinal axonal boutons in the mouse dLGN all showed that much higher RGC convergence is possible, with up to 90 RGCs from multiple classes innervating a single dLGN neuron. In their recent preprint, Román Rosón et al. also address these questions.
Key findings
The authors examine connectivity between the retina and the thalamus by first defining the overall diversity of RGC classes targeting the dLGN. Using a combination of mouse genetics and viral manipulations in the dLGN, a genetically encoded calcium indicator was selectively expressed in RGCs projecting to the dLGN. Two-photon calcium imaging was then used to measure the responses of labelled RGCs to a standardized set of stimuli.
On the basis of their response properties, the labelled dLGN-projecting RGCs were then assigned to previously characterised functional RGC classes (Baden et al., 2016). This showed that 75% of all RGC types target the dLGN, and that certain classes are over- or under-represented within the dLGN projecting population.
To look into how this wealth of information is processed by the thalamus, the authors were interested in the degree of functional RGC convergence onto single TC neurons in the dLGN. Here, functional convergence is defined as the number of different RGC types that meaningfully drive the responses of a TC cell to visual stimuli.
To this end, they first performed extracellular single-unit recordings of geniculate neurons in head-fixed mice, which were presented the same set of stimuli as the retinas. From this data, they observed that both TC and RGC responses showed similar diversity.
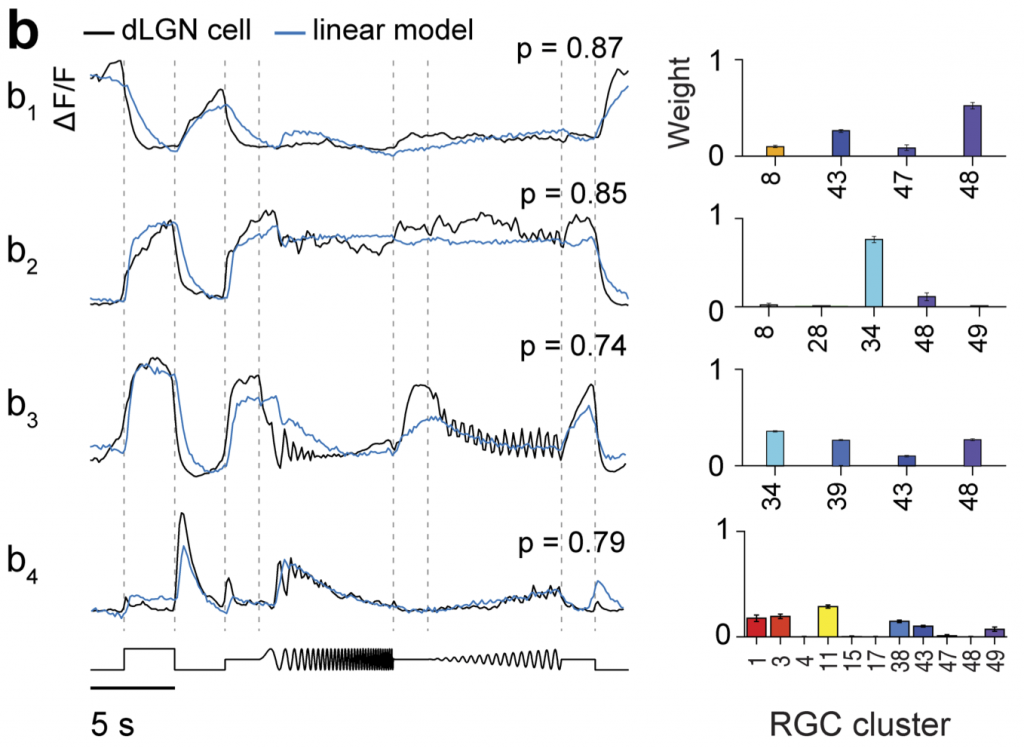
Finally, the study used a linear model to predict dLGN responses as a weighted sum of excitatory RGC inputs. This modelling implies that on average just 2 different RGC groups drive the response of a dLGN TC neuron. The estimate, however, varied depending on what threshold was chosen for inputs to be considered functionally relevant. Taken together, this would therefore suggest a limited functional retinal convergence in the visual thalamus.
Interestingly, the model also showed that RGC classes overrepresented in the dLGN projecting RGC population contributed significantly to dLGN TC neuron responses. This would suggest that the information these RGCs relay is particularly significant for subsequent visual computations.
What I like about this work
I think this work addresses the question of the relation between structure and function with an elegant and comprehensive experimental design, which combines viral-based anatomical tracing with calcium imaging. To my knowledge it is also the most complete functional characterisation of RGC classes that target the dLGN.
At a structural level, connectivity between the RGCs and dLGN TC neurons appears complex and ‘fuzzy’, as reported by many studies, with evidence of high RGC convergence onto some thalamic targets. The results here however suggest that this structural complexity does not necessarily translate functionally, as only few RGC types seem to dominate the functional responses of TC neurons. Therefore, this preprint could provide an explanation for opposing findings in the field, arrived at by using very different methodologies (e.g. electrophysiology vs electron microscopy and rabies virus tracing).
One caveat, however, is that the resolution is low. For example, tracing from the dLGN is done at a population level, with no distinction between excitatory and inhibitory dLGN neurons. Below, I also discuss some considerations that may require experiments at a finer scale, or using more complex visual paradigms, to address.
Further questions
As also pointed out by the authors, in addition to TC cells, local circuits in the dLGN include inhibitory interneurons, and projections from the visual cortex, the reticular nucleus, midbrain and brainstem nuclei. RGCs are known to also target dLGN interneurons. It remains largely unknown how all of these local circuit features affect TC responses to visual inputs, especially with regards to these recent findings on structural and functional organization of retinogeniculate connectivity.
Additionally, in mice (and even more prominently in carnivores and primates), the dLGN is composed of parallel visual pathways. For example there is a significant distinction between its shell and core regions, in terms of the inputs they receive and their projection patterns in the cortex. It would thus be interesting to look if there are any differences in functional RGC convergence between different dLGN microcircuits.
References
Baden, T. et al. (2016) The functional diversity of retinal ganglion cells in the mouse. Nature, 529, 345-50.
Sign up to customise the site to your preferences and to receive alerts
Register hereAlso in the neuroscience category:
Generalization and extinction of learned fear alter primary sensory input to the brain
Kathleen Ngo et al.
Fear conditioning biases olfactory sensory neuron frequencies across generations
Yuwei Wu et al.
Chromatin priming and co-factor availability shape lineage response to the neuronal pioneer factor ASCL1 in pluripotency
Reinier Prosee, Jethro Lundie-Brown
preLists in the neuroscience category:
preLighters’ choice – Handpicked DevBio preprints
preLighters with expertise across developmental and stem cell biology have nominated a few developmental biology (and related) preprints they’re excited about and explain in a few paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Theodora Stougiannou et al. |
BSDB Spring Meeting: Molecules to Morphogenesis
The British Society for Developmental Biology (BSDB) Spring Meeting Molecules to Morphogenesis was held from 23–26 March 2026 at the University of Warwick (UK). This meeting brought together a vibrant community of researchers to discuss how molecular mechanisms are integrated across scales to drive morphogenesis, spanning diverse model systems and approaches. This preList contains preprints by presenters from the talk and poster sessions at the meeting. Please do get in touch at preLights@biologists.com if you notice any relevant preprints that we may have missed.
| List by | Ingrid Tsang |
Keystone Symposium on Stem Cell Models in Embryology 2026
The Keystone Symposium on Stem Cell Models in Embryology, 2026, was organised by Jun Wu (UT Southwestern), Jianping Fu (University of Michigan) and Miki Ebisuya (TU Dresden) and held at Asilomar Conference Grounds in California (US). The meeting discussed recent advances made in establishing stem-cell-based embryo models, what fundamental insights into developmental processes have been gleaned from them, as well as how they are beginning to be applied more widely. This prelist contains preprints by presenters at the talk and poster sessions at the conference, which our Reviews Editor in attendance spotted. Please do reach out to preLights@biologists.com if you notice any that we’ve missed.
| List by | Ingrid Tsang |
November in preprints – DevBio & Stem cell biology
preLighters with expertise across developmental and stem cell biology have nominated a few developmental and stem cell biology (and related) preprints posted in November they’re excited about and explain in a single paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Aline Grata et al. |
October in preprints – DevBio & Stem cell biology
Each month, preLighters with expertise across developmental and stem cell biology nominate a few recent developmental and stem cell biology (and related) preprints they’re excited about and explain in a single paragraph why. Short, snappy picks from working scientists — a quick way to spot fresh ideas, bold methods and papers worth reading in full. These preprints can all be found in the October preprint list published on the Node.
| List by | Deevitha Balasubramanian et al. |
October in preprints – Cell biology edition
Different preLighters, with expertise across cell biology, have worked together to create this preprint reading list for researchers with an interest in cell biology. This month, most picks fall under (1) Cell organelles and organisation, followed by (2) Mechanosignaling and mechanotransduction, (3) Cell cycle and division and (4) Cell migration
| List by | Matthew Davies et al. |
July in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: (1) Cell Signalling and Mechanosensing (2) Cell Cycle and Division (3) Cell Migration and Cytoskeleton (4) Cancer Biology (5) Cell Organelles and Organisation
| List by | Girish Kale et al. |
May in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) Biochemistry/metabolism 2) Cancer cell Biology 3) Cell adhesion, migration and cytoskeleton 4) Cell organelles and organisation 5) Cell signalling and 6) Genetics
| List by | Barbora Knotkova et al. |
April in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) biochemistry/metabolism 2) cell cycle and division 3) cell organelles and organisation 4) cell signalling and mechanosensing 5) (epi)genetics
| List by | Vibha SINGH et al. |
Biologists @ 100 conference preList
This preList aims to capture all preprints being discussed at the Biologists @100 conference in Liverpool, UK, either as part of the poster sessions or the (flash/short/full-length) talks.
| List by | Reinier Prosee, Jonathan Townson |
2024 Hypothalamus GRC
This 2024 Hypothalamus GRC (Gordon Research Conference) preList offers an overview of cutting-edge research focused on the hypothalamus, a critical brain region involved in regulating homeostasis, behavior, and neuroendocrine functions. The studies included cover a range of topics, including neural circuits, molecular mechanisms, and the role of the hypothalamus in health and disease. This collection highlights some of the latest advances in understanding hypothalamic function, with potential implications for treating disorders such as obesity, stress, and metabolic diseases.
| List by | Nathalie Krauth |
‘In preprints’ from Development 2022-2023
A list of the preprints featured in Development's 'In preprints' articles between 2022-2023
| List by | Alex Eve, Katherine Brown |
CSHL 87th Symposium: Stem Cells
Preprints mentioned by speakers at the #CSHLsymp23
| List by | Alex Eve |
Journal of Cell Science meeting ‘Imaging Cell Dynamics’
This preList highlights the preprints discussed at the JCS meeting 'Imaging Cell Dynamics'. The meeting was held from 14 - 17 May 2023 in Lisbon, Portugal and was organised by Erika Holzbaur, Jennifer Lippincott-Schwartz, Rob Parton and Michael Way.
| List by | Helen Zenner |
FENS 2020
A collection of preprints presented during the virtual meeting of the Federation of European Neuroscience Societies (FENS) in 2020
| List by | Ana Dorrego-Rivas |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
SDB 78th Annual Meeting 2019
A curation of the preprints presented at the SDB meeting in Boston, July 26-30 2019. The preList will be updated throughout the duration of the meeting.
| List by | Alex Eve |
Autophagy
Preprints on autophagy and lysosomal degradation and its role in neurodegeneration and disease. Includes molecular mechanisms, upstream signalling and regulation as well as studies on pharmaceutical interventions to upregulate the process.
| List by | Sandra Malmgren Hill |
Young Embryologist Network Conference 2019
Preprints presented at the Young Embryologist Network 2019 conference, 13 May, The Francis Crick Institute, London
| List by | Alex Eve |











 (No Ratings Yet)
(No Ratings Yet)