Optical Control of G-Actin with a Photoswitchable Latrunculin
Posted on: 14 September 2023 , updated on: 10 May 2024
Preprint posted on 19 July 2023
Article now published in Journal of the American Chemical Society at http://dx.doi.org/10.1021/jacs.3c10776
Switch on, switch off. @n_vep and colleagues (@RuiyangB, @OertnerLab, @DirkTrauner) describe photoswitchable actin destabilisers activated by blue light. Preprint highlighted by @zhanghe_goh
Selected by Zhang-He GohCategories: pharmacology and toxicology
Updated 11 May 2024 with a postLight by Zhang-He Goh
This highlighted preprint has now been published in the Journal of the American Chemical Society.
In this work, Vepřek and co-workers from the Trauner group report the development of a series of photoswitchable G-actin stabilisers based on the latrunculin family. Generally, these probes are activated when exposed to light irradiation, enabling them to bind to G-actin monomers reversibly.
The main difference between the preprint and the published version seems to be a set of experiments involving OptoLat, the authors’ best probe. In these pyrene-actin polymerisation assays, the authors showed that OptoLat exhibited light-dependent inhibition of polymerisation that decays with time.
Congratulations again to the authors.
Background of the preprint
Actin is an important structural protein in eukaryotic cells. Due to its biological importance, its homeostasis is tightly regulated by multiple biological mechanisms. This provides many opportunities by which actin can be modulated—several small molecule modulators of actin have been discovered. However, the complexity of these processes also makes the study of actin very challenging.
As a result of highly collaborative efforts led by the Trauner group and the Arndt group, a series of photoswitchable versions of the F-actin stabiliser jasplakinolide, called optojasps, were created that stabilise F-actin precisely and reversibly.1, 2 In this work, Vepřek and co-workers (from the Trauner group) develop a series of photoswitchable G-actin stabilisers based on the latrunculins. Consisting of a diazene linker, these probes are usually in the inactive trans- position; when exposed to light irradiation, they then convert to the active cis- position (Figure 1). Due to their ability to photoswitch, these probes can bind to G-actin monomers reversibly when light is shone on them. The authors could thus use these probes to control G-actin dynamics in a space- and time-dependent manner. First, they performed a series of assays to identify the most useful probe, which they named OptoLat. They then tested OptoLat in various cell lines and in more complex tissues.
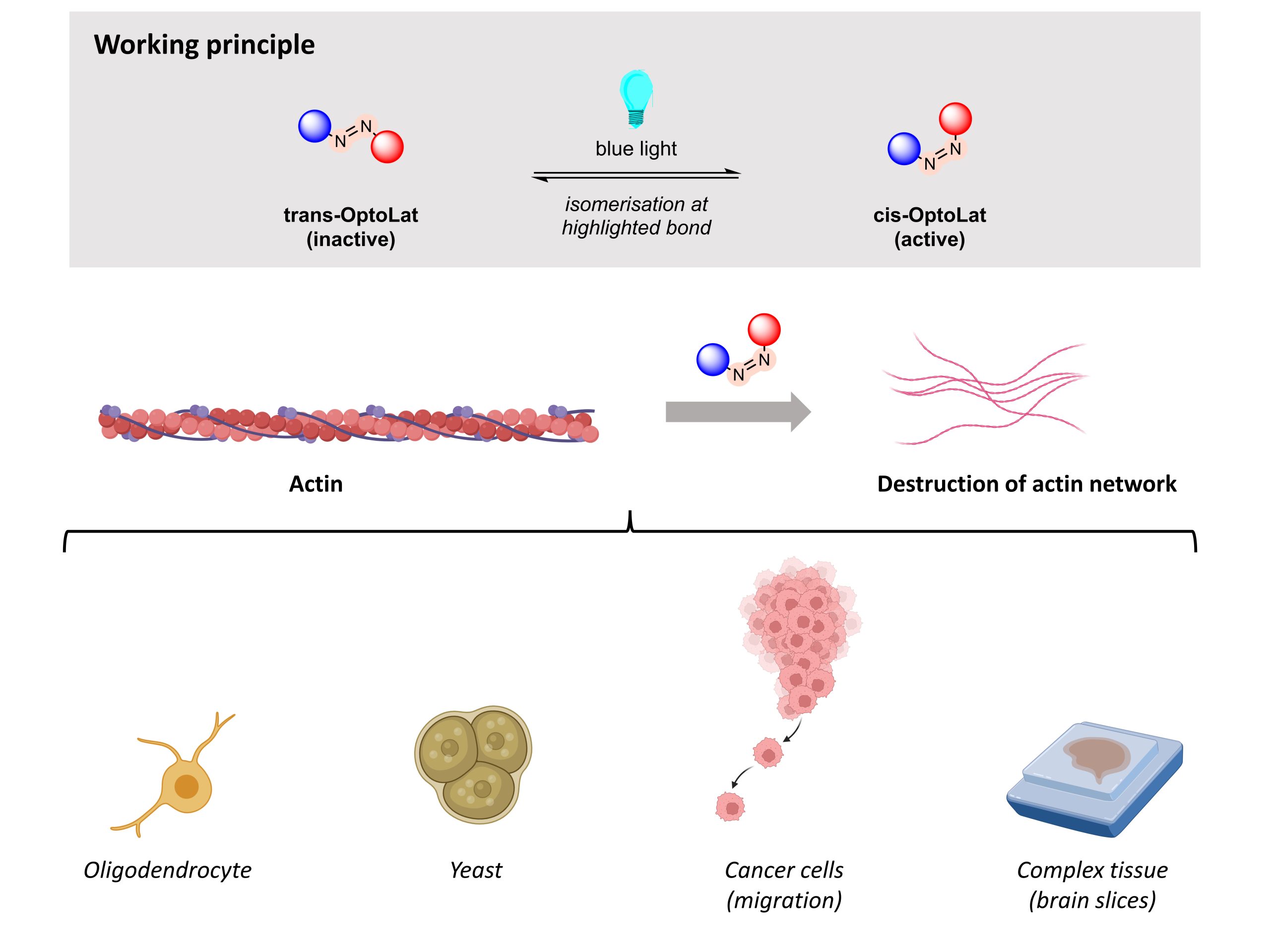
Figure 1. Overview of the work reported by Vepřek and co-workers in their preprint.
Key findings of this preprint
Vepřek and co-workers first set about designing and synthesising their latrunculin photochemical probes based on the structure-activity relationship of known latrunculin binders. Broadly, these latrunculin analogues either bore macrocyclic lactones or ester linkages to the azobenzene and diazocine groups. The authors also made two more structural changes to their latrunculin probes. First, they modified the vector around the aromatic diazene connection to modulate the steric clash between their probes and the protein following photoisomerisation of the diazene bond. Second, they replaced the ester linkages with aryl ethers to promote metabolic stability and to maximise steric effects when the probes bind to the protein pocket.
Among these probes, the authors identified one that exhibited the best photophysical properties, i.e. the most red-shifted absorption spectrum and the fastest thermal relaxation, which they named OptoLat. In further tests, they also confirmed that photoswitching was fully reversible (i.e. could undergo complete deactivation and reactivation) and persisted for at least 30 cycles. In light-dependent cell proliferation assays, OptoLat was activated by irradiation with blue light, with an EC50 in the micromolar range.
Next, Vepřek and co-workers tested OptoLat in cell-based assays to probe actin dynamics and their effects on cellular structure and function. In oligodendrocytes, the authors proved that OptoLat’s antiproliferative effect was caused by its ability to destabilise actin networks upon irradiation with blue light.
With this in hand, the authors investigated OptoLat’s effects in various biological systems. Actin plays important roles in yeast cellular division and proliferation and cell migration and the authors found that exposing OptoLat-treated yeast to blue light led to a significant decrease in actin cables within 5 minutes. Similarly, OptoLat reduced the migration of invasive cancer cells in a light-dependent fashion, though the concentration of OptoLat used under these conditions was not cytotoxic. Further experiments by the authors showed that OptoLat activation in live cells destroyed actin networks and that these changes were fully reversible.
Finally, the authors tested OptoLat in a more complex tissue—the brain. In these experiments, the authors monitored the cell shape and motility of microglia following OptoLat treatment and activation. They found that OptoLat activation did not affect cell shape, but significantly reduced motility; changes in motility were reversed after the OptoLat-treated cells were no longer exposed to light.
What I like about this preprint
Photoswitchable probes have many uses in chemical biology: they are often used to investigate cellular organisation and dynamics. Here, Vepřek and co-workers outline their development of a light-activated actin de-stabiliser, OptoLat. I think this preprint is interesting for two reasons.
First, the biological applications will be broad. Actin is a highly-conserved structural protein, so probes that target actin will be applicable in almost all organisms. However, selectivity will be essential for specific control. This problem is partly resolved by OptoLat, because it is only activated in the presence of blue light. Vepřek and co-workers could thus control when OptoLat was activated, the duration of the activation, and the regions by which it was activated.
Second, I find the chemistry of photoswitchable probes rather fascinating. Specifically, to be able to interconvert the cis- and trans- isomers of OptoLat readily, reversibly, and in a repeated manner without degradation of the probe is quite challenging because these light-sensitive probes can degrade each time they are excited by light. Moreover, the authors describe a simple route to synthesise OptoLat of just about 10 steps in their preprint. Synthetic simplicity is important in these projects because it directly affects synthetic chemists’ throughput in making a variety of structures to understand the structure-activity relationship of these probes and identify the best one. While one of the synthetic steps in this route produces a pair of diastereomers, they can be separately purified and might anyway be useful intermediates that could lead to the further exploration of diastereomeric analogues.
Future directions
Future work around the development of OptoLat and its analogues will probably involve looking into their potential biological uses. Open questions prompting future research for instance include: are these diazene structures orthogonal to other functional handles? That is, could we install different functional groups onto these probes, and selectively activate them by shining different wavelengths of light? For example, diazirines are useful photochemical probes in protein labelling. In theory, it would thus be possible to develop probes in which shining light of a certain wavelength would change its shape, and shining light of a different wavelength would allow it to perform another function, such as labelling. These developments would conceivably broaden the applications of these probes.
Acknowledgements
Images created using Microsoft Powerpoint, ChemDraw, and BioRender.
Questions for authors
- Did thermal relaxation times differ among the probes and were these differences significant?
References
- M. Borowiak, F. Küllmer, F. Gegenfurtner, S. Peil, V. Nasufovic, S. Zahler, O. Thorn-Seshold, D. Trauner and H.-D. Arndt, Journal of the American Chemical Society, 2020, 142, 9240-9249.
- F. Küllmer, N. A. Vepřek, M. Borowiak, V. Nasufović, S. Barutzki, O. Thorn‐Seshold, H. D. Arndt and D. Trauner, Angewandte Chemie International Edition, 2022, 61.
doi: https://doi.org/10.1242/prelights.35576
Read preprintSign up to customise the site to your preferences and to receive alerts
Register hereAlso in the pharmacology and toxicology category:
The Endocannabinoid System’s Contribution to Placebo Analgesia
Thomas Nicodemo Arrieta et al.
Small Molecule Agonists of TREM2 Reprogram Microglia and Protect Synapses in Human Alzheimer’s Models
Dina Kabbara
Snake venom metalloproteinases are predominantly responsible for the cytotoxic effects of certain African viper venoms
Daniel Osorno Valencia
preLists in the pharmacology and toxicology category:
SciELO preprints – From 2025 onwards
SciELO has become a cornerstone of open, multilingual scholarly communication across Latin America. Its preprint server, SciELO preprints, is expanding the global reach of preprinted research from the region (for more information, see our interview with Carolina Tanigushi). This preList brings together biological, English language SciELO preprints to help readers discover emerging work from the Global South. By highlighting these preprints in one place, we aim to support visibility, encourage early feedback, and showcase the vibrant research communities contributing to SciELO’s open science ecosystem.
| List by | Carolina Tanigushi |
April in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) biochemistry/metabolism 2) cell cycle and division 3) cell organelles and organisation 4) cell signalling and mechanosensing 5) (epi)genetics
| List by | Vibha SINGH et al. |
FENS 2020
A collection of preprints presented during the virtual meeting of the Federation of European Neuroscience Societies (FENS) in 2020
| List by | Ana Dorrego-Rivas |
COVID-19 / SARS-CoV-2 preprints
List of important preprints dealing with the ongoing coronavirus outbreak. See http://covidpreprints.com for additional resources and timeline, and https://connect.biorxiv.org/relate/content/181 for full list of bioRxiv and medRxiv preprints on this topic
| List by | Dey Lab, Zhang-He Goh |
1
Drug use in special populations
Any drugs that are being used in special populations: Patients with liver and kidney failure, in paediatrics, in geriatrics, and in pregnant or lactating patients. Includes the discovery of factors that could potentially affect drug use in these special populations.
| List by | Zhang-He Goh |
Toxicology of toxicants, existing therapeutics, and investigational drugs
Preprints that describe the toxicology of environmental pollutants and existing and upcoming drugs. Includes both toxicokinetics and toxicodynamics, as well as technological improvements that will help in the characterisation of this field.
| List by | Zhang-He Goh |
Antimicrobials: Discovery, clinical use, and development of resistance
Preprints that describe the discovery of new antimicrobials and any improvements made regarding their clinical use. Includes preprints that detail the factors affecting antimicrobial selection and the development of antimicrobial resistance.
| List by | Zhang-He Goh |
Anticancer agents: Discovery and clinical use
Preprints that describe the discovery of anticancer agents and their clinical use. Includes both small molecules and macromolecules like biologics.
| List by | Zhang-He Goh |
Advances in Drug Delivery
Advances in formulation technology or targeted delivery methods that describe or develop the distribution of small molecules or large macromolecules to specific parts of the body.
| List by | Zhang-He Goh |











 (No Ratings Yet)
(No Ratings Yet)