PPARδ activation in microglia drives a transcriptional response that primes phagocytic function while countering inflammatory activation
Posted on: 11 February 2026 , updated on: 16 February 2026
Preprint posted on 19 December 2025
PPAR excellence: microglial transcription factor shows potential as a therapeutic target for Alzheimer’s disease
Selected by Isabel PaineCategories: neuroscience
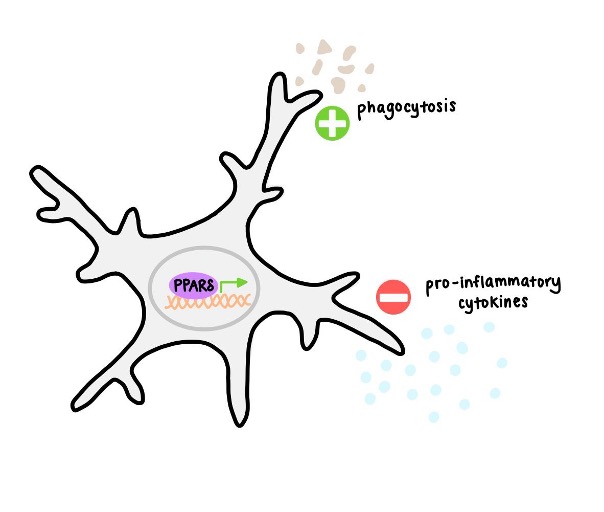
Introduction/Background:
As the resident immune cells of the central nervous system, microglia serve a protective role in the normal functioning of the brain. However, microglia have also been shown to contribute to the progression of neurodegenerative disease, such as Alzheimer’s disease (AD). The characterization of microglia in these disease contexts is needed, as it is becoming more clear that microglia can exhibit many different functional states– meaning they can take on various roles or behaviors– based on their morphology and gene expression profile.
PPARδ is a member of the PPAR transcription factor family that is highly expressed by microglia. PPARs are lipid sensors and mediate inflammation. Agonists for PPARδ specifically have been tested as treatments for Huntington’s disease (HD) and sporadic AD. Although PPARδ has shown potential in this regard, it is still not entirely clear how it modifies the function of microglia. The authors performed several different experiments to establish the effects of PPARδ activation on microglia.
Why I highlight this work:
Microglia identity is an area of research that has evolved extensively over recent years, transitioning from the idea of dichotomic categories to the understanding that a spectrum of microglia states exists. Research on microglia phenotypes in the context of various diseases has always been interesting to me, especially because I study cellular changes in the brain during the process of aging and in vascular diseases. I found that this study thoroughly investigates the role of the transcription factor PPARδ, and presents it as a promising therapeutic target in microglia for AD.
Key Findings:
PPARδ agonism in mice increases markers of phagocytosis while decreasing markers of inflammation and migration
In their initial in vivo experiment, mice treated with a PPARδ agonist showed significantly up- and down-regulated genes in bulk RNA sequencing. From gene ontology analysis, they determined that many down-regulated genes were overall related to immune activation and cell migration, suggesting that these processes lessen. Looking at specific regulated genes that were significantly regulated also demonstrated an increase in phagocytosis and decrease in neuroinflammation.
PPARδ agonism reduces inflammation and migration in vitro
In a complementary in vitro study, the authors differentiated induced pluripotent stem cells (iPSCs) into microglia. They treated the resulting microglia with lipopolysaccharide (LPS) to induce an inflammatory response. Microglia that were co-treated with PPARδ exhibited a decrease in the release of inflammatory cytokines. A migration assay with the cultured cells also revealed less migration in microglia pre-treated with PPARδ, consistent with their in vivo RNA sequencing results. They also found that PPARδ agonist-treated microglia showed increased phagocytosis of fluorescently labelled rat synaptosomes and amyloid beta.
Additionally, the authors used a novel approach to assess the microglial response to a broader neurodegenerative injury. Induced microglia exposed to “brain powder” in combination with PPARδ agonist showed significant and opposing transcriptome differences compared to those treated with PPARδ antagonist. Again, genes upregulated with PPARδ agonism were associated with decreased inflammatory pathways and migration and increased lipid metabolism.
PPARδ interacts with and modulates PU.1 in vitro
The authors took a closer look at the relationship between PPARδ and the transcription factor PU.1, which regulates many of the inflammatory genes they found to be downregulated by PPARδ. Co-expression and co-immunoprecipitation revealed an interaction between the two proteins. Further, PPARδ agonist reduced the interaction between PPARδ and PU.1 and the activity of PU.1 in vitro.
Inflammatory markers decrease with PPARδ agonism in HD and tauopathy mice
Bringing in more clinical relevance, the authors returned to an in vivo system to examine how PPARδ agonism affects inflammation in both a HD and tauopathy mouse model. In both models, as well as in wildtype mice, they found a decrease in inflammatory genes upon treatment with PPARδ agonist.
Questions and Future Directions:
- Do you have any idea whether there may be proteins that could interact with PPARδ to mediate the changed expression of genes related to other processes of interest, such as migration, lipid processing, and phagocytosis?
- Have you done in vitro experiments with microglia co-cultured with neurons and other glial cells? What differences would you expect to observe when including these other cell types?
- Given your results looking at phagocytosis of AD-relevant substrates, do you plan to look at PPARδ agonism in an amyloid beta mouse model?
doi: https://doi.org/10.1242/prelights.42921
Read preprintSign up to customise the site to your preferences and to receive alerts
Register hereAlso in the neuroscience category:
Consciously detecting and recognizing a past visual word after its sensory trace is gone
Maitri Manjunath
Exogenous Amyloid Sequences: Their Role in Amyloid-Beta Heterotypic Aggregation
Cyntia Alves Conceição, Marcus Oliveira
Human Assembloid Model of Emergent Neurotropic Enteroviruses
Mitchell Sarmie
preLists in the neuroscience category:
BSDB Spring Meeting: Molecules to Morphogenesis
The British Society for Developmental Biology (BSDB) Spring Meeting Molecules to Morphogenesis was held from 23–26 March 2026 at the University of Warwick (UK). This meeting brought together a vibrant community of researchers to discuss how molecular mechanisms are integrated across scales to drive morphogenesis, spanning diverse model systems and approaches. This preList contains preprints by presenters from the talk and poster sessions at the meeting. Please do get in touch at preLights@biologists.com if you notice any relevant preprints that we may have missed.
| List by | Ingrid Tsang |
Keystone Symposium on Stem Cell Models in Embryology 2026
The Keystone Symposium on Stem Cell Models in Embryology, 2026, was organised by Jun Wu (UT Southwestern), Jianping Fu (University of Michigan) and Miki Ebisuya (TU Dresden) and held at Asilomar Conference Grounds in California (US). The meeting discussed recent advances made in establishing stem-cell-based embryo models, what fundamental insights into developmental processes have been gleaned from them, as well as how they are beginning to be applied more widely. This prelist contains preprints by presenters at the talk and poster sessions at the conference, which our Reviews Editor in attendance spotted. Please do reach out to preLights@biologists.com if you notice any that we’ve missed.
| List by | Ingrid Tsang |
November in preprints – DevBio & Stem cell biology
preLighters with expertise across developmental and stem cell biology have nominated a few developmental and stem cell biology (and related) preprints posted in November they’re excited about and explain in a single paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Aline Grata et al. |
October in preprints – DevBio & Stem cell biology
Each month, preLighters with expertise across developmental and stem cell biology nominate a few recent developmental and stem cell biology (and related) preprints they’re excited about and explain in a single paragraph why. Short, snappy picks from working scientists — a quick way to spot fresh ideas, bold methods and papers worth reading in full. These preprints can all be found in the October preprint list published on the Node.
| List by | Deevitha Balasubramanian et al. |
October in preprints – Cell biology edition
Different preLighters, with expertise across cell biology, have worked together to create this preprint reading list for researchers with an interest in cell biology. This month, most picks fall under (1) Cell organelles and organisation, followed by (2) Mechanosignaling and mechanotransduction, (3) Cell cycle and division and (4) Cell migration
| List by | Matthew Davies et al. |
July in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: (1) Cell Signalling and Mechanosensing (2) Cell Cycle and Division (3) Cell Migration and Cytoskeleton (4) Cancer Biology (5) Cell Organelles and Organisation
| List by | Girish Kale et al. |
May in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) Biochemistry/metabolism 2) Cancer cell Biology 3) Cell adhesion, migration and cytoskeleton 4) Cell organelles and organisation 5) Cell signalling and 6) Genetics
| List by | Barbora Knotkova et al. |
April in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) biochemistry/metabolism 2) cell cycle and division 3) cell organelles and organisation 4) cell signalling and mechanosensing 5) (epi)genetics
| List by | Vibha SINGH et al. |
Biologists @ 100 conference preList
This preList aims to capture all preprints being discussed at the Biologists @100 conference in Liverpool, UK, either as part of the poster sessions or the (flash/short/full-length) talks.
| List by | Reinier Prosee, Jonathan Townson |
2024 Hypothalamus GRC
This 2024 Hypothalamus GRC (Gordon Research Conference) preList offers an overview of cutting-edge research focused on the hypothalamus, a critical brain region involved in regulating homeostasis, behavior, and neuroendocrine functions. The studies included cover a range of topics, including neural circuits, molecular mechanisms, and the role of the hypothalamus in health and disease. This collection highlights some of the latest advances in understanding hypothalamic function, with potential implications for treating disorders such as obesity, stress, and metabolic diseases.
| List by | Nathalie Krauth |
‘In preprints’ from Development 2022-2023
A list of the preprints featured in Development's 'In preprints' articles between 2022-2023
| List by | Alex Eve, Katherine Brown |
CSHL 87th Symposium: Stem Cells
Preprints mentioned by speakers at the #CSHLsymp23
| List by | Alex Eve |
Journal of Cell Science meeting ‘Imaging Cell Dynamics’
This preList highlights the preprints discussed at the JCS meeting 'Imaging Cell Dynamics'. The meeting was held from 14 - 17 May 2023 in Lisbon, Portugal and was organised by Erika Holzbaur, Jennifer Lippincott-Schwartz, Rob Parton and Michael Way.
| List by | Helen Zenner |
FENS 2020
A collection of preprints presented during the virtual meeting of the Federation of European Neuroscience Societies (FENS) in 2020
| List by | Ana Dorrego-Rivas |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
SDB 78th Annual Meeting 2019
A curation of the preprints presented at the SDB meeting in Boston, July 26-30 2019. The preList will be updated throughout the duration of the meeting.
| List by | Alex Eve |
Autophagy
Preprints on autophagy and lysosomal degradation and its role in neurodegeneration and disease. Includes molecular mechanisms, upstream signalling and regulation as well as studies on pharmaceutical interventions to upregulate the process.
| List by | Sandra Malmgren Hill |
Young Embryologist Network Conference 2019
Preprints presented at the Young Embryologist Network 2019 conference, 13 May, The Francis Crick Institute, London
| List by | Alex Eve |











 (No Ratings Yet)
(No Ratings Yet)