Miniaturized Devices for Bioluminescence Imaging in Freely Behaving Animals
Posted on: 11 July 2020
Preprint posted on 16 June 2020
Categories: bioengineering, neuroscience
Background
Microscopic imaging of fluorescent indicators to track neural activity is an increasingly important tool in systems neuroscience.Since almost a decade, fluorescence miniature microscopy in vivo has been a major advance, enabling single-cell resolution imaging in freely behaving animals. Since its release it has been used to study questions in domains ranging from memory representation to olfactory processing. Some limitations of fluorescence-based microscopy include photobleaching and phototoxicity; and that the signal-to-noise ratio for localizing discrete signals in the tissue is limited, due to autofluorescence from unintended sources, and non-homogeneous illumination due to the scattering of excitation light. Bioluminescence provides an alternative kind of activity-dependent light indicator, whereby light is generated via chemiluminescent reaction between luciferase (an enzyme) and luciferin (a substrate). Bioluminescent cells do not suffer from photobleaching, phototoxicity, or excitation scattering, and these point sources do not create autofluorescence. Further, bioluminescent indicators also do not require excitation light optics: the removal of this component should make lighter and lower cost microscope with fewer assembly parts. While there has been progress in making brighter and faster bioluminescence indicators, parallel advances in imaging hardware have not followed. In their work, Celinskis et al (1) investigate whether fluorescent miniature microscopes can be rendered sensitive enough to detect bioluminescence, and tested its capacity for detecting bioluminescent signals in vitro and in vivo.
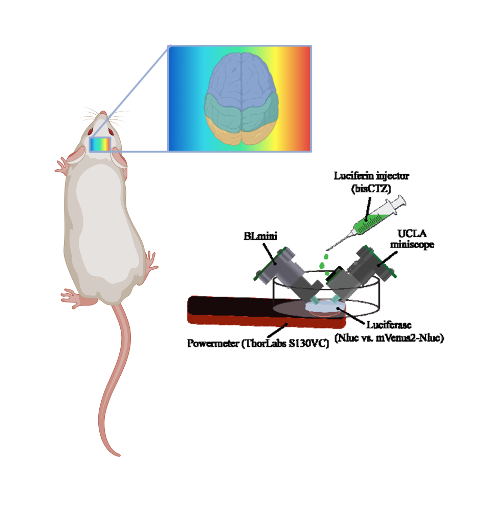
Key findings and developments
In this work, Celinskis et al modified the miniscope v3.2 ((2), referred to as the UCLA miniscope) to create the Bioluminescence Miniscope (BLmini) removing the components necessary for excitation light optics, allowing simplification of the microscope’s architecture. They configured parameters including power setup, acquisition speed, gain, and exposure time. They then performed an in vivo comparison of the UCLA miniscope, comparing signals emanating from a mouse expressing mNeonGreen tethered to EkL9H luciferase (i.e. a shrimp-based luciferase variant also referred to as NCS2). Both miniscopes were able to detect bioluminescence from both constructs, but the BLmini showed significantly stronger signal. Other aspects compared included mass, number of components, cost, and power consumption. BLmini is 22% lighter in weight, has 45% fewer components, is up to 58% less expensive, offers up to 15 times stronger signal (as dichroic filtering is not required) and is sensitive enough to capture spatiotemporal dynamics of bioluminescence in the brain with a signal-to-noise ratio of 34 dB. The BLmini was also able to capture the temporal dynamics of in vivo bioluminescence. A limitation discussed is that as BLmini does not rely on any form of cooling, it consequently suffers from significantly stronger thermoelectric background noise.
This progress represents a key step in the refinement of this approach, which can prove transformative to an important imaging approach.
What I like about this preprint
I think this is an important methodological advance with the potential for multiple applications in many research fields. As the authors discussed, the miniscope was revolutionary, and so far nothing similar had been produced for bioluminescence. The authors provide that opportunity now, which offers complementary advantages to fluorescence imaging.
Open questions
- You used for testing, EkL9H. What is the range of wavelengths that the BLmini can detect? Would the signal-to-noise ratio become limiting for probes such as NanoLuc, Renilla Luciferase, or Gaussia Luciferase?
- Is there a chance to do dual-bioluminescence imaging?
- Since you used the miniscope as a basis, is there a chance to have a platform that uses both fluorescence and bioluminescence, without much increased cost or weight, in order to use the advantages of both imaging modalities?
- Is it only possible to use the BLmini in the brain of free-moving animals? Has it been attempted in the spinal cord or other organs accessible dorsally?
- Are there any specific limitations to bioluminescence imaging associated with free- movement?
References
- Celinskis D et al, Miniaturized devices for bioluminescence imaging in freely behaving animals, bioRxiv 2020.
- Cai DJ et al, A shared neural ensemble links distinct contextual memories encoded close in time, Nature, 2016
doi: https://doi.org/10.1242/prelights.22918
Read preprintSign up to customise the site to your preferences and to receive alerts
Register hereAlso in the bioengineering category:
Detergent-Triggered Membrane Remodelling Monitored via Intramembrane Fluorescence De-Quenching
Cyntia Alves Conceição, Marcus Oliveira
A Novel Chimeric Antigen Receptor (CAR) - Strategy to Target EGFRVIII-Mutated Glioblastoma Cells via Macrophages
Dina Kabbara
Human pluripotent stem cell-derived macrophages modify development of human kidney organoids
Theodora Stougiannou
Also in the neuroscience category:
Generalization and extinction of learned fear alter primary sensory input to the brain
Kathleen Ngo et al.
Fear conditioning biases olfactory sensory neuron frequencies across generations
Yuwei Wu et al.
Chromatin priming and co-factor availability shape lineage response to the neuronal pioneer factor ASCL1 in pluripotency
Reinier Prosee, Jethro Lundie-Brown
preLists in the bioengineering category:
October in preprints – DevBio & Stem cell biology
Each month, preLighters with expertise across developmental and stem cell biology nominate a few recent developmental and stem cell biology (and related) preprints they’re excited about and explain in a single paragraph why. Short, snappy picks from working scientists — a quick way to spot fresh ideas, bold methods and papers worth reading in full. These preprints can all be found in the October preprint list published on the Node.
| List by | Deevitha Balasubramanian et al. |
CSHL 87th Symposium: Stem Cells
Preprints mentioned by speakers at the #CSHLsymp23
| List by | Alex Eve |
EMBL Synthetic Morphogenesis: From Gene Circuits to Tissue Architecture (2021)
A list of preprints mentioned at the #EESmorphoG virtual meeting in 2021.
| List by | Alex Eve |
3D Gastruloids
A curated list of preprints related to Gastruloids (in vitro models of early development obtained by 3D aggregation of embryonic cells). Updated until July 2021.
| List by | Paul Gerald L. Sanchez and Stefano Vianello |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
EMBL Seeing is Believing – Imaging the Molecular Processes of Life
Preprints discussed at the 2019 edition of Seeing is Believing, at EMBL Heidelberg from the 9th-12th October 2019
| List by | Dey Lab |
Lung Disease and Regeneration
This preprint list compiles highlights from the field of lung biology.
| List by | Rob Hynds |
Advances in microscopy
This preList highlights exciting unpublished preprint articles describing advances in microscopy with a focus on light-sheet microscopy.
| List by | Stephan Daetwyler |
Also in the neuroscience category:
preLighters’ choice – Handpicked DevBio preprints
preLighters with expertise across developmental and stem cell biology have nominated a few developmental biology (and related) preprints they’re excited about and explain in a few paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Theodora Stougiannou et al. |
BSDB Spring Meeting: Molecules to Morphogenesis
The British Society for Developmental Biology (BSDB) Spring Meeting Molecules to Morphogenesis was held from 23–26 March 2026 at the University of Warwick (UK). This meeting brought together a vibrant community of researchers to discuss how molecular mechanisms are integrated across scales to drive morphogenesis, spanning diverse model systems and approaches. This preList contains preprints by presenters from the talk and poster sessions at the meeting. Please do get in touch at preLights@biologists.com if you notice any relevant preprints that we may have missed.
| List by | Ingrid Tsang |
Keystone Symposium on Stem Cell Models in Embryology 2026
The Keystone Symposium on Stem Cell Models in Embryology, 2026, was organised by Jun Wu (UT Southwestern), Jianping Fu (University of Michigan) and Miki Ebisuya (TU Dresden) and held at Asilomar Conference Grounds in California (US). The meeting discussed recent advances made in establishing stem-cell-based embryo models, what fundamental insights into developmental processes have been gleaned from them, as well as how they are beginning to be applied more widely. This prelist contains preprints by presenters at the talk and poster sessions at the conference, which our Reviews Editor in attendance spotted. Please do reach out to preLights@biologists.com if you notice any that we’ve missed.
| List by | Ingrid Tsang |
November in preprints – DevBio & Stem cell biology
preLighters with expertise across developmental and stem cell biology have nominated a few developmental and stem cell biology (and related) preprints posted in November they’re excited about and explain in a single paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Aline Grata et al. |
October in preprints – DevBio & Stem cell biology
Each month, preLighters with expertise across developmental and stem cell biology nominate a few recent developmental and stem cell biology (and related) preprints they’re excited about and explain in a single paragraph why. Short, snappy picks from working scientists — a quick way to spot fresh ideas, bold methods and papers worth reading in full. These preprints can all be found in the October preprint list published on the Node.
| List by | Deevitha Balasubramanian et al. |
October in preprints – Cell biology edition
Different preLighters, with expertise across cell biology, have worked together to create this preprint reading list for researchers with an interest in cell biology. This month, most picks fall under (1) Cell organelles and organisation, followed by (2) Mechanosignaling and mechanotransduction, (3) Cell cycle and division and (4) Cell migration
| List by | Matthew Davies et al. |
July in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: (1) Cell Signalling and Mechanosensing (2) Cell Cycle and Division (3) Cell Migration and Cytoskeleton (4) Cancer Biology (5) Cell Organelles and Organisation
| List by | Girish Kale et al. |
May in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) Biochemistry/metabolism 2) Cancer cell Biology 3) Cell adhesion, migration and cytoskeleton 4) Cell organelles and organisation 5) Cell signalling and 6) Genetics
| List by | Barbora Knotkova et al. |
April in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) biochemistry/metabolism 2) cell cycle and division 3) cell organelles and organisation 4) cell signalling and mechanosensing 5) (epi)genetics
| List by | Vibha SINGH et al. |
Biologists @ 100 conference preList
This preList aims to capture all preprints being discussed at the Biologists @100 conference in Liverpool, UK, either as part of the poster sessions or the (flash/short/full-length) talks.
| List by | Reinier Prosee, Jonathan Townson |
2024 Hypothalamus GRC
This 2024 Hypothalamus GRC (Gordon Research Conference) preList offers an overview of cutting-edge research focused on the hypothalamus, a critical brain region involved in regulating homeostasis, behavior, and neuroendocrine functions. The studies included cover a range of topics, including neural circuits, molecular mechanisms, and the role of the hypothalamus in health and disease. This collection highlights some of the latest advances in understanding hypothalamic function, with potential implications for treating disorders such as obesity, stress, and metabolic diseases.
| List by | Nathalie Krauth |
‘In preprints’ from Development 2022-2023
A list of the preprints featured in Development's 'In preprints' articles between 2022-2023
| List by | Alex Eve, Katherine Brown |
CSHL 87th Symposium: Stem Cells
Preprints mentioned by speakers at the #CSHLsymp23
| List by | Alex Eve |
Journal of Cell Science meeting ‘Imaging Cell Dynamics’
This preList highlights the preprints discussed at the JCS meeting 'Imaging Cell Dynamics'. The meeting was held from 14 - 17 May 2023 in Lisbon, Portugal and was organised by Erika Holzbaur, Jennifer Lippincott-Schwartz, Rob Parton and Michael Way.
| List by | Helen Zenner |
FENS 2020
A collection of preprints presented during the virtual meeting of the Federation of European Neuroscience Societies (FENS) in 2020
| List by | Ana Dorrego-Rivas |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
SDB 78th Annual Meeting 2019
A curation of the preprints presented at the SDB meeting in Boston, July 26-30 2019. The preList will be updated throughout the duration of the meeting.
| List by | Alex Eve |
Autophagy
Preprints on autophagy and lysosomal degradation and its role in neurodegeneration and disease. Includes molecular mechanisms, upstream signalling and regulation as well as studies on pharmaceutical interventions to upregulate the process.
| List by | Sandra Malmgren Hill |
Young Embryologist Network Conference 2019
Preprints presented at the Young Embryologist Network 2019 conference, 13 May, The Francis Crick Institute, London
| List by | Alex Eve |











 (No Ratings Yet)
(No Ratings Yet)