Novel insights in the pathophysiology of α-synuclein dysregulation on D2 receptor activity contributing to the vulnerability of dopamine neurons
Posted on: 26 May 2021 , updated on: 27 May 2021
Preprint posted on 30 March 2021
Preventing neuronal vulnerability in PD: Activation of D2 receptors rescues survival and complexity of alpha-Synuclein overexpressing dopaminergic neurones.
Selected by Emma WilsonCategories: neuroscience, physiology
Background
Parkinson’s disease (PD) is the second most common neurodegenerative disorder and is characterised by the loss of dopaminergic neurones and proteinous inclusions known as Lewy bodies in the ventral midbrain. These neurones play a vital role in the initiation and control of movement meaning that patients often present with rigidity, tremor, and slowness of movement and inability to initiate movement. It is known that damage or loss of function of these neurones precedes their death1.
Lewy bodies are proteinous accumulations of primarily alpha-Synuclein. The gene that encodes for alpha-Synuclein, SNCA, is known to be mutated, duplicated or triplicated in autosomal dominant forms of PD. There is also evidence that overexpression of alpha-Synuclein plays a role in idiopathic PD. Overexpression of wild type (WT) alpha-Synuclein is sufficient to alter the pathophysiology of dopaminergic neurones resulting in their vulnerability and eventual loss. The exact mechanism for this is not yet known.
In this preprint, the authors used primary mouse cultures of dopaminergic neurones in conjunction with adeno virus expressing WT human alpha-Synuclein under a dopaminergic promoter (Tyrosine Hydroxylase (TH)). These neurones were compared with control non alpha-Synuclein expressing cells. They saw high specificity of alpha-Synuclein in TH positive/Dopaminergic neurones (Figure 1).
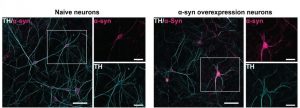
Figure 1: Immunofluorescence images showing expression and co-localisation of TH and -Synuclein in naïve and -Synuclein overexpression dopaminergic neuron model. Adapted from Figure 1C in the preprint under a CC-BY 4.0 licence.
Key findings
The overexpression of alpha-Synuclein disrupts Dopamine (D2) receptor autoinhibition resulting in altered calcium signalling and increased firing rate of mouse dopaminergic neurones.
It is known that loss of function precedes neurone loss in PD1. The authors wanted to investigate if the overexpression of WT human alpha-Synuclein in mouse dopaminergic neurones altered their electrical activity.
Following overexpression of alpha-Synuclein the width and amplitude of the Ca2+ signal increased, which was complemented by an increase in neuronal firing recorded by whole cell current recordings. This suggests that overexpression of alpha-Synuclein can increase firing rate of dopaminergic neurones.
Both firing activity and Ca2+ dynamics are tightly regulated by D2 autoinhibitory receptor which suppresses electrical activity of dopaminergic neurones upon dopamine binding2,3. The D2 receptor agonist, Quinpirole, was utilised to show that those cells expressing alpha-Synuclein maintained their Ca2+ signal and firing rate unlike control neurones which saw an inhibition of both Ca2+ signals and firing rate when treated with D2 agonist. Furthermore, blocking D2 receptors, in control neurones mirrored the effects of firing rate as seen with alpha-Synuclein overexpression. These data suggests that -Synuclein impacts D2 receptors to alter Ca2+ signalling and firing rate of mouse DA neurones.
The overexpression of alpha-Synuclein decreases the complexity and survival of Dopaminergic neurones.
Dopaminergic neurones are neuronally complex, have large branching neurites and projections fields. In PD patients there is a decrease in complexity of dopaminergic neurones4. The authors investigated the complexity and survival of mouse dopaminergic neurones overexpressing alpha-Synuclein. To do this they use Sholl analysis, which examines neurite projection area and field, number of intersections (branching) and circularity, alongside Tyrosine Hydroxylase (TH) immunofluorescence which marks dopaminergic neurones (Figure 2a). In neurones overexpressing alpha-Synuclein there was a decrease in neuronal processes, projection area and circularity accompanied by a decreased in the amount of TH expressing cells (Figure 2 (b,c). This suggests that neurones expressing alpha-Synuclein are less complex and may be informative about the progression of alpha-Synuclein pathology prior to neuronal loss.

Figure 2: (a) Schematic illustrating the measurable parameters of dopaminergic neurone. (b,c,d) schematic representations of naïve, those with overexpressed alpha-Synuclein and D2 agonist pre-treatment. Adapted from Figure 6 in the preprint under a CC-BY 4.0 licence
Pharmacological activation of D2 receptors could be a potential target to alleviate alpha-Synuclein pathology.
The authors had previously found an unexpected effect of prolonged exposure of dopaminergic neurones to the agonist Quinone. They saw that this rescued the firing properties to the alpha-Synuclein expressing neurones to that of the control. They further investigated other key defects seen in these alpha-Synuclein expressing neurones such as: neuron complexity, survival, Calcium dynamics, spontaneous firing and dopamine release and synthesis. They found partial rescue of these factors with prolonged exposure (48h) of neurones to Quinone. This highlights D2 receptor agonists as potential drug targets and an area for further investigation (Figure 2)
Importance of this work/ How it moves to field forwards
This paper highlights D2 receptors as a potential target that could be used to alleviate alpha-Synuclein pathology in a range of diseases including PD and Lewy body dementia. With an aging population it is increasingly important to highlight a diverse range of treatments. What is especially interesting is that any treatments involving D2 dopamine receptor could be given preventatively treating the cause of disease rather than the symptoms. However, early diagnosis of neurodegenerative disease is still difficult. I specifically liked the use of many complementary methods to solidify their finding in addition to examining the functionality of the neurone not just survival.
Open Questions
- Was this aggregated alpha-Synuclein? Could this have a different effect?
- What do you think the effects of mutated alpha-Synuclein on these parameters such as firing rate and the impact that D2 agonists could have? Have you thought about investigating this?
- This is conducted in mouse cells, are there plans for this be conducted using human model? IPSC’s?
- Are there any drugs which target D2 receptors, that have already been approved for other uses which could be tested in your model and potentially re-purposed?
doi: https://doi.org/10.1242/prelights.29164
Read preprintSign up to customise the site to your preferences and to receive alerts
Register hereAlso in the neuroscience category:
Gut microbiome changes over the course of multiple sclerosis differentially influence autoimmune neuroinflammation
Carole Djagang et al.
Prenatal Stress Differentially Shapes Adult Behavior in Male and Female Offspring
Alice Gennevois et al.
Consciously detecting and recognizing a past visual word after its sensory trace is gone
Maitri Manjunath
Also in the physiology category:
Resilience to cardiac aging in Greenland shark Somniosus microcephalus
Theodora Stougiannou
Trade-offs between surviving and thriving: A careful balance of physiological limitations and reproductive effort under thermal stress
Tshepiso Majelantle
Imaging cellular activity simultaneously across all organs of a vertebrate reveals body-wide circuits
Muhammed Sinan Malik
preLists in the neuroscience category:
preLighters’ choice – Handpicked DevBio preprints
preLighters with expertise across developmental and stem cell biology have nominated a few developmental biology (and related) preprints they’re excited about and explain in a few paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Theodora Stougiannou et al. |
BSDB Spring Meeting: Molecules to Morphogenesis
The British Society for Developmental Biology (BSDB) Spring Meeting Molecules to Morphogenesis was held from 23–26 March 2026 at the University of Warwick (UK). This meeting brought together a vibrant community of researchers to discuss how molecular mechanisms are integrated across scales to drive morphogenesis, spanning diverse model systems and approaches. This preList contains preprints by presenters from the talk and poster sessions at the meeting. Please do get in touch at preLights@biologists.com if you notice any relevant preprints that we may have missed.
| List by | Ingrid Tsang |
Keystone Symposium on Stem Cell Models in Embryology 2026
The Keystone Symposium on Stem Cell Models in Embryology, 2026, was organised by Jun Wu (UT Southwestern), Jianping Fu (University of Michigan) and Miki Ebisuya (TU Dresden) and held at Asilomar Conference Grounds in California (US). The meeting discussed recent advances made in establishing stem-cell-based embryo models, what fundamental insights into developmental processes have been gleaned from them, as well as how they are beginning to be applied more widely. This prelist contains preprints by presenters at the talk and poster sessions at the conference, which our Reviews Editor in attendance spotted. Please do reach out to preLights@biologists.com if you notice any that we’ve missed.
| List by | Ingrid Tsang |
November in preprints – DevBio & Stem cell biology
preLighters with expertise across developmental and stem cell biology have nominated a few developmental and stem cell biology (and related) preprints posted in November they’re excited about and explain in a single paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Aline Grata et al. |
October in preprints – DevBio & Stem cell biology
Each month, preLighters with expertise across developmental and stem cell biology nominate a few recent developmental and stem cell biology (and related) preprints they’re excited about and explain in a single paragraph why. Short, snappy picks from working scientists — a quick way to spot fresh ideas, bold methods and papers worth reading in full. These preprints can all be found in the October preprint list published on the Node.
| List by | Deevitha Balasubramanian et al. |
October in preprints – Cell biology edition
Different preLighters, with expertise across cell biology, have worked together to create this preprint reading list for researchers with an interest in cell biology. This month, most picks fall under (1) Cell organelles and organisation, followed by (2) Mechanosignaling and mechanotransduction, (3) Cell cycle and division and (4) Cell migration
| List by | Matthew Davies et al. |
July in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: (1) Cell Signalling and Mechanosensing (2) Cell Cycle and Division (3) Cell Migration and Cytoskeleton (4) Cancer Biology (5) Cell Organelles and Organisation
| List by | Girish Kale et al. |
May in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) Biochemistry/metabolism 2) Cancer cell Biology 3) Cell adhesion, migration and cytoskeleton 4) Cell organelles and organisation 5) Cell signalling and 6) Genetics
| List by | Barbora Knotkova et al. |
April in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) biochemistry/metabolism 2) cell cycle and division 3) cell organelles and organisation 4) cell signalling and mechanosensing 5) (epi)genetics
| List by | Vibha SINGH et al. |
Biologists @ 100 conference preList
This preList aims to capture all preprints being discussed at the Biologists @100 conference in Liverpool, UK, either as part of the poster sessions or the (flash/short/full-length) talks.
| List by | Reinier Prosee, Jonathan Townson |
2024 Hypothalamus GRC
This 2024 Hypothalamus GRC (Gordon Research Conference) preList offers an overview of cutting-edge research focused on the hypothalamus, a critical brain region involved in regulating homeostasis, behavior, and neuroendocrine functions. The studies included cover a range of topics, including neural circuits, molecular mechanisms, and the role of the hypothalamus in health and disease. This collection highlights some of the latest advances in understanding hypothalamic function, with potential implications for treating disorders such as obesity, stress, and metabolic diseases.
| List by | Nathalie Krauth |
‘In preprints’ from Development 2022-2023
A list of the preprints featured in Development's 'In preprints' articles between 2022-2023
| List by | Alex Eve, Katherine Brown |
CSHL 87th Symposium: Stem Cells
Preprints mentioned by speakers at the #CSHLsymp23
| List by | Alex Eve |
Journal of Cell Science meeting ‘Imaging Cell Dynamics’
This preList highlights the preprints discussed at the JCS meeting 'Imaging Cell Dynamics'. The meeting was held from 14 - 17 May 2023 in Lisbon, Portugal and was organised by Erika Holzbaur, Jennifer Lippincott-Schwartz, Rob Parton and Michael Way.
| List by | Helen Zenner |
FENS 2020
A collection of preprints presented during the virtual meeting of the Federation of European Neuroscience Societies (FENS) in 2020
| List by | Ana Dorrego-Rivas |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
SDB 78th Annual Meeting 2019
A curation of the preprints presented at the SDB meeting in Boston, July 26-30 2019. The preList will be updated throughout the duration of the meeting.
| List by | Alex Eve |
Autophagy
Preprints on autophagy and lysosomal degradation and its role in neurodegeneration and disease. Includes molecular mechanisms, upstream signalling and regulation as well as studies on pharmaceutical interventions to upregulate the process.
| List by | Sandra Malmgren Hill |
Young Embryologist Network Conference 2019
Preprints presented at the Young Embryologist Network 2019 conference, 13 May, The Francis Crick Institute, London
| List by | Alex Eve |
Also in the physiology category:
preLighters’ choice – Handpicked DevBio preprints
preLighters with expertise across developmental and stem cell biology have nominated a few developmental biology (and related) preprints they’re excited about and explain in a few paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Theodora Stougiannou et al. |
Keystone Symposium on Stem Cell Models in Embryology 2026
The Keystone Symposium on Stem Cell Models in Embryology, 2026, was organised by Jun Wu (UT Southwestern), Jianping Fu (University of Michigan) and Miki Ebisuya (TU Dresden) and held at Asilomar Conference Grounds in California (US). The meeting discussed recent advances made in establishing stem-cell-based embryo models, what fundamental insights into developmental processes have been gleaned from them, as well as how they are beginning to be applied more widely. This prelist contains preprints by presenters at the talk and poster sessions at the conference, which our Reviews Editor in attendance spotted. Please do reach out to preLights@biologists.com if you notice any that we’ve missed.
| List by | Ingrid Tsang |
October in preprints – DevBio & Stem cell biology
Each month, preLighters with expertise across developmental and stem cell biology nominate a few recent developmental and stem cell biology (and related) preprints they’re excited about and explain in a single paragraph why. Short, snappy picks from working scientists — a quick way to spot fresh ideas, bold methods and papers worth reading in full. These preprints can all be found in the October preprint list published on the Node.
| List by | Deevitha Balasubramanian et al. |
Biologists @ 100 conference preList
This preList aims to capture all preprints being discussed at the Biologists @100 conference in Liverpool, UK, either as part of the poster sessions or the (flash/short/full-length) talks.
| List by | Reinier Prosee, Jonathan Townson |
Fibroblasts
The advances in fibroblast biology preList explores the recent discoveries and preprints of the fibroblast world. Get ready to immerse yourself with this list created for fibroblasts aficionados and lovers, and beyond. Here, my goal is to include preprints of fibroblast biology, heterogeneity, fate, extracellular matrix, behavior, topography, single-cell atlases, spatial transcriptomics, and their matrix!
| List by | Osvaldo Contreras |
FENS 2020
A collection of preprints presented during the virtual meeting of the Federation of European Neuroscience Societies (FENS) in 2020
| List by | Ana Dorrego-Rivas |
TAGC 2020
Preprints recently presented at the virtual Allied Genetics Conference, April 22-26, 2020. #TAGC20
| List by | Maiko Kitaoka et al. |
Autophagy
Preprints on autophagy and lysosomal degradation and its role in neurodegeneration and disease. Includes molecular mechanisms, upstream signalling and regulation as well as studies on pharmaceutical interventions to upregulate the process.
| List by | Sandra Malmgren Hill |
Cellular metabolism
A curated list of preprints related to cellular metabolism at Biorxiv by Pablo Ranea Robles from the Prelights community. Special interest on lipid metabolism, peroxisomes and mitochondria.
| List by | Pablo Ranea Robles |











 (No Ratings Yet)
(No Ratings Yet)