Species-specific developmental timing is associated with global differences in protein stability in mouse and human
Posted on: 16 January 2020 , updated on: 17 January 2020
Preprint posted on 31 December 2019
Do you want (your embryonic development) to go slower? Make your proteins live longer!
Selected by Irepan Salvador-MartinezCategories: developmental biology
Introduction
“Diese Verschiebung kann sowohl den Teile Ort, als die Zeit der Erscheinung betreffen.
Jene erstere nennen wir Heterotopie, diese letztere Heterochronie”
(“This shift can affect both the location and the time of appearance.
We call the former heterotopy, the latter heterochrony.”)
Haeckel, 1875
Ernst Haeckel coined the term “heterochrony” to describe temporal deviations from his infamous recapitulation theory or biogenetic law. Its modern use, however, refers to any evolutionary change in developmental timing [1]. Changes in developmental timing between species are readily evident and have fascinated embryologists for decades (if not centuries). Before the genomic era, heterochrony was perceived as the temporal shift in the appearance of a morphological trait, when comparing embryos of different species (e.g. the appearance of the heart between human and fish). Now it is known that the underlying gene regulatory machinery to form homologous structures is greatly conserved between species, and heterochronic shifts can be recognised by comparing the temporal dynamics of gene expression through the development of such structures. The mechanisms responsible for these shifts have remained elusive, however.
About the preprint
Rayon and collaborators investigated the temporal variation in the differentiation of motor neurons (MNs) between mouse and human. MNs develop from progenitors in the embryonic spinal cord after being exposed to Sonic Hedgehog emanating from the notochord. In both species, MN progenitors can be identified by the expression of the genes Olig2 and Nkx6.1 due to the conservation of its Gene Regulatory Network (GRN). Although the temporal sequence of gene expression is conserved in both species, its rate is different. In mouse, MN differentiation takes 3-4 days while in human it takes ~2 weeks. The authors decided to use the term “developmental allochrony” instead of heterochrony, to highlight that in this case there is a scaling of developmental timing between these species without any apparent morphological trait alteration (see Author’s response below).
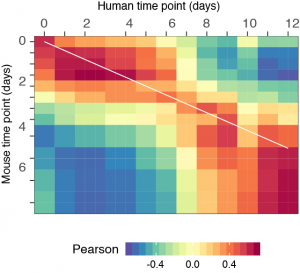
To investigate what could be causing this timing difference they decided to use MN differentiation in vitro for both species. First, they confirmed that their in vitro system could recapitulate the species-specific differentiation timing, assayed by the expression of marker genes such as Olig2 and Nkx6.1. Then they showed, by comparing bulk transcriptomes, that this difference in developmental tempo was present on a global scale as a 2.5 fold decrease in the gene expression rate in human compared to mouse (see Figure 1). So what could be causing this global change in the differentiation of MNs? The authors elegantly performed a series of tests to try to identify the culprit.
First, they tested if the slower pace in human could be explained by a reduced response to Shh signalling. To do this they increased the amount of Shh signaling in human MN progenitors (via addition of smoothened agonists). The results showed that species-specific sensitivity to Shh is not responsible for the difference in developmental tempo. Secondly, they tested if species-specific differences in the genetic sequence of Olig2 (the major regulator of MN progenitors) could explain the global tempo shift. For this, they used a mouse ESC line that contains the arm of chromosome 21 where the gene Olig2 is found. They found that these cells containing the human version of Olig2 had the same expression dynamics of cells with the mouse version of it, indicating that the temporal control of gene expression depends on the cellular environment and not on inter-specific differences in the genetic sequence of Oligo2 or its cis-regulatory regions.
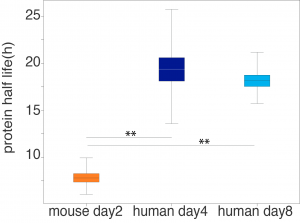
Finally, Rayon and collaborators set out to determine if the degradation rate of transcripts and proteins could be involved in the temporal shift of MN differentiation. An assay of global mRNA stability showed no differences between mouse and human neural progenitors. A protein stability assay showed, however, that the half-life of the proteome in mouse neural progenitors was shorter than in human progenitors, by approximately 2.5 fold (Figure 2).
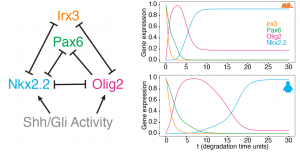
They used a mathematical model of the MN GRN, previously developed by the same group, to test if increasing the stability of transcription factors could account for the observed temporal differences. Indeed, the simulations showed that increasing the stability of the TF genes present in the GRN resulted in a slower pace of gene expression sequence, similar to the experimentally observed one (Figure 3 ). Given that the cell cycle also shows a similar temporal difference between species, the authors suggest that differences in cell cycle rate between mouse and human cells could also be a consequence of a global change in protein stability.
A previous analysis [2] demonstrated that the speed of the biochemical reactions (including protein degradation) involved in the intracellular network that drives gene expression oscillation in the presomitic mesoderm could explain the 2.5 fold timing difference of the segmentation clocks between the mouse and human (see prelights [3] and [4]). The results of Rayon and collaborators show that the timing differences between mouse and human motor neuron differentiation is linked to differences in protein stability between these species, suggesting that protein stability could regulate developmental timings at a global scale. How the protein stability can be regulated at a global level remains to be determined.
Questions to the authors:
1. Did you come up with the term “developmental allochrony”? I could not find any previous usage of it.
2. The GRN of MN differentiation consists of TF mutual inhibitions. Do you think this GRN structure is especially sensitive to protein stability? Could other type of GRNs (e.g. a more hierarchical GRN) be affected less by protein stability?
3. The idea of protein stability affecting both developmental timing and cell cycle rate is attractive as it could act as a mechanism to couple patterning with cell cycle dynamics during development. Do you think this could have evolved as a scaling mechanism of developmental timing, or is it an intrinsic property of developmental systems?
References
[1] Gould SJ. The Uses of Heterochrony. In: McKinney ML, ed. Heterochrony in Evolution: A Multidisciplinary Approach. Boston, MA: Springer US; 1988:1-13. doi:10.1007/978-1-4899-0795-0_1
[2] Matsuda M, Hayashi H, Garcia-Ojalvo J, et al. Species-specific oscillation periods of human and mouse segmentation clocks are due to cell autonomous differences in biochemical reaction parameters. bioRxiv. 2019. https://www.biorxiv.org/content/10.1101/650648v1.
doi: https://doi.org/10.1242/prelights.16298
Read preprintSign up to customise the site to your preferences and to receive alerts
Register hereAlso in the developmental biology category:
Tissue mechanics and systemic signaling safeguard epithelial tissue against spindle misorientation
Ruoheng Li
Human pluripotent stem cell-derived macrophages modify development of human kidney organoids
Theodora Stougiannou
Junctional Heterogeneity Shapes Epithelial Morphospace
Bhaval Parmar
preLists in the developmental biology category:
November in preprints – DevBio & Stem cell biology
preLighters with expertise across developmental and stem cell biology have nominated a few developmental and stem cell biology (and related) preprints posted in November they’re excited about and explain in a single paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Aline Grata et al. |
October in preprints – DevBio & Stem cell biology
Each month, preLighters with expertise across developmental and stem cell biology nominate a few recent developmental and stem cell biology (and related) preprints they’re excited about and explain in a single paragraph why. Short, snappy picks from working scientists — a quick way to spot fresh ideas, bold methods and papers worth reading in full. These preprints can all be found in the October preprint list published on the Node.
| List by | Deevitha Balasubramanian et al. |
October in preprints – Cell biology edition
Different preLighters, with expertise across cell biology, have worked together to create this preprint reading list for researchers with an interest in cell biology. This month, most picks fall under (1) Cell organelles and organisation, followed by (2) Mechanosignaling and mechanotransduction, (3) Cell cycle and division and (4) Cell migration
| List by | Matthew Davies et al. |
June in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: (1) Cell organelles and organisation (2) Cell signaling and mechanosensation (3) Genetics/gene expression (4) Biochemistry (5) Cytoskeleton
| List by | Barbora Knotkova et al. |
Keystone Symposium – Metabolic and Nutritional Control of Development and Cell Fate
This preList contains preprints discussed during the Metabolic and Nutritional Control of Development and Cell Fate Keystone Symposia. This conference was organized by Lydia Finley and Ralph J. DeBerardinis and held in the Wylie Center and Tupper Manor at Endicott College, Beverly, MA, United States from May 7th to 9th 2025. This meeting marked the first in-person gathering of leading researchers exploring how metabolism influences development, including processes like cell fate, tissue patterning, and organ function, through nutrient availability and metabolic regulation. By integrating modern metabolic tools with genetic and epidemiological insights across model organisms, this event highlighted key mechanisms and identified open questions to advance the emerging field of developmental metabolism.
| List by | Virginia Savy, Martin Estermann |
Biologists @ 100 conference preList
This preList aims to capture all preprints being discussed at the Biologists @100 conference in Liverpool, UK, either as part of the poster sessions or the (flash/short/full-length) talks.
| List by | Reinier Prosee, Jonathan Townson |
BSDB/GenSoc Spring Meeting 2024
A list of preprints highlighted at the British Society for Developmental Biology and Genetics Society joint Spring meeting 2024 at Warwick, UK.
| List by | Joyce Yu, Katherine Brown |
GfE/ DSDB meeting 2024
This preList highlights the preprints discussed at the 2024 joint German and Dutch developmental biology societies meeting that took place in March 2024 in Osnabrück, Germany.
| List by | Joyce Yu |
‘In preprints’ from Development 2022-2023
A list of the preprints featured in Development's 'In preprints' articles between 2022-2023
| List by | Alex Eve, Katherine Brown |
preLights peer support – preprints of interest
This is a preprint repository to organise the preprints and preLights covered through the 'preLights peer support' initiative.
| List by | preLights peer support |
The Society for Developmental Biology 82nd Annual Meeting
This preList is made up of the preprints discussed during the Society for Developmental Biology 82nd Annual Meeting that took place in Chicago in July 2023.
| List by | Joyce Yu, Katherine Brown |
CSHL 87th Symposium: Stem Cells
Preprints mentioned by speakers at the #CSHLsymp23
| List by | Alex Eve |
Journal of Cell Science meeting ‘Imaging Cell Dynamics’
This preList highlights the preprints discussed at the JCS meeting 'Imaging Cell Dynamics'. The meeting was held from 14 - 17 May 2023 in Lisbon, Portugal and was organised by Erika Holzbaur, Jennifer Lippincott-Schwartz, Rob Parton and Michael Way.
| List by | Helen Zenner |
9th International Symposium on the Biology of Vertebrate Sex Determination
This preList contains preprints discussed during the 9th International Symposium on the Biology of Vertebrate Sex Determination. This conference was held in Kona, Hawaii from April 17th to 21st 2023.
| List by | Martin Estermann |
Alumni picks – preLights 5th Birthday
This preList contains preprints that were picked and highlighted by preLights Alumni - an initiative that was set up to mark preLights 5th birthday. More entries will follow throughout February and March 2023.
| List by | Sergio Menchero et al. |
CellBio 2022 – An ASCB/EMBO Meeting
This preLists features preprints that were discussed and presented during the CellBio 2022 meeting in Washington, DC in December 2022.
| List by | Nadja Hümpfer et al. |
2nd Conference of the Visegrád Group Society for Developmental Biology
Preprints from the 2nd Conference of the Visegrád Group Society for Developmental Biology (2-5 September, 2021, Szeged, Hungary)
| List by | Nándor Lipták |
Fibroblasts
The advances in fibroblast biology preList explores the recent discoveries and preprints of the fibroblast world. Get ready to immerse yourself with this list created for fibroblasts aficionados and lovers, and beyond. Here, my goal is to include preprints of fibroblast biology, heterogeneity, fate, extracellular matrix, behavior, topography, single-cell atlases, spatial transcriptomics, and their matrix!
| List by | Osvaldo Contreras |
EMBL Synthetic Morphogenesis: From Gene Circuits to Tissue Architecture (2021)
A list of preprints mentioned at the #EESmorphoG virtual meeting in 2021.
| List by | Alex Eve |
EMBL Conference: From functional genomics to systems biology
Preprints presented at the virtual EMBL conference "from functional genomics and systems biology", 16-19 November 2020
| List by | Jesus Victorino |
Single Cell Biology 2020
A list of preprints mentioned at the Wellcome Genome Campus Single Cell Biology 2020 meeting.
| List by | Alex Eve |
Society for Developmental Biology 79th Annual Meeting
Preprints at SDB 2020
| List by | Irepan Salvador-Martinez, Martin Estermann |
FENS 2020
A collection of preprints presented during the virtual meeting of the Federation of European Neuroscience Societies (FENS) in 2020
| List by | Ana Dorrego-Rivas |
Planar Cell Polarity – PCP
This preList contains preprints about the latest findings on Planar Cell Polarity (PCP) in various model organisms at the molecular, cellular and tissue levels.
| List by | Ana Dorrego-Rivas |
Cell Polarity
Recent research from the field of cell polarity is summarized in this list of preprints. It comprises of studies focusing on various forms of cell polarity ranging from epithelial polarity, planar cell polarity to front-to-rear polarity.
| List by | Yamini Ravichandran |
TAGC 2020
Preprints recently presented at the virtual Allied Genetics Conference, April 22-26, 2020. #TAGC20
| List by | Maiko Kitaoka et al. |
3D Gastruloids
A curated list of preprints related to Gastruloids (in vitro models of early development obtained by 3D aggregation of embryonic cells). Updated until July 2021.
| List by | Paul Gerald L. Sanchez and Stefano Vianello |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
EDBC Alicante 2019
Preprints presented at the European Developmental Biology Congress (EDBC) in Alicante, October 23-26 2019.
| List by | Sergio Menchero et al. |
EMBL Seeing is Believing – Imaging the Molecular Processes of Life
Preprints discussed at the 2019 edition of Seeing is Believing, at EMBL Heidelberg from the 9th-12th October 2019
| List by | Dey Lab |
SDB 78th Annual Meeting 2019
A curation of the preprints presented at the SDB meeting in Boston, July 26-30 2019. The preList will be updated throughout the duration of the meeting.
| List by | Alex Eve |
Lung Disease and Regeneration
This preprint list compiles highlights from the field of lung biology.
| List by | Rob Hynds |
Young Embryologist Network Conference 2019
Preprints presented at the Young Embryologist Network 2019 conference, 13 May, The Francis Crick Institute, London
| List by | Alex Eve |
Pattern formation during development
The aim of this preList is to integrate results about the mechanisms that govern patterning during development, from genes implicated in the processes to theoritical models of pattern formation in nature.
| List by | Alexa Sadier |
BSCB/BSDB Annual Meeting 2019
Preprints presented at the BSCB/BSDB Annual Meeting 2019
| List by | Dey Lab |
Zebrafish immunology
A compilation of cutting-edge research that uses the zebrafish as a model system to elucidate novel immunological mechanisms in health and disease.
| List by | Shikha Nayar |











 (No Ratings Yet)
(No Ratings Yet)