Analysis of the role of Nidogen/entactin in basement membrane assembly and morphogenesis in Drosophila
Posted on: 30 August 2018 , updated on: 31 August 2018
Preprint posted on 13 June 2018
Article now published in PLOS Genetics at http://dx.doi.org/10.1371/journal.pgen.1007483
Necessary, or unnecessary, that is the question: Nidogen is not essential for overall Drosophila morphogenesis and viability, but required for proper basement membrane assembly in certain tissues.
Selected by Nargess KhalilgharibiCategories: developmental biology
Context and background:
Basement membrane (BM) is a specific form of the extracellular matrix lining the basal side of the epithelial and endothelial tissues. The main BM constituents (i.e. laminin, collagen IV, perlecan and nidogen) have been conserved across evolution. The proper composition and structure of basement membrane is essential for correct tissue growth, organisation, differentiation and maintenance. Different BM components play distinct roles in its assembly and maintenance: it has been shown that laminin is required for incorporation of collagen IV into the BM, which will then help determine tissue shape, first by imposing a mechanical constriction on the cells and second through recruitment of perlecan that counters the constrictive effect of collagen IV1. However, the role of nidogen in BM assembly and maintenance is still not well understood. Indeed, in vitro studies propose that nidogen acts as a linker between laminin and collagen IV networks, thus playing a key role in BM assembly. On the other hand, in vivo experiments have shown that loss of the only nidogen gene in C. elegans2 or either of the two nidogen genes in mice3, 4 does not affect BM assembly, organogenesis or survival, while simultaneous loss of the two genes in mice have shown to be lethal due to tissue-specific BM defects5. The authors of this preprint have investigated the role of nidogen in BM assembly of different tissues, using Drosophila as a model system.
Key findings:
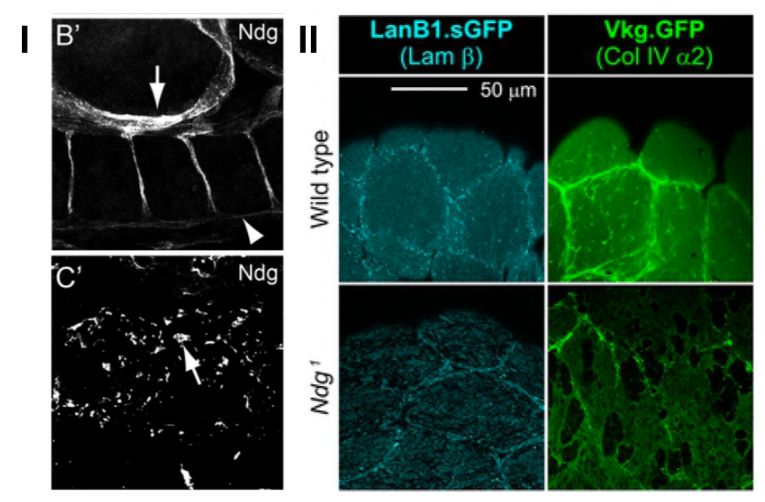
Invertebrates, including Drosophila have only one nidogen gene. Developing a new anti-nidogen antibody, Dai et al. show that nidogen is found in the BM of most embryonic, larval and adult tissues. Interestingly, they also show that depending on the type of embryonic tissue, nidogen is assembled in different patterns. For example, while nidogen is present in continuous linear structures around muscles, gut and the ventral nerve cord (VNC), it has a dotted pattern along the visceral mesoderm (Fig. I). The authors identify fat body and blood cells as the main sources of nidogen, although some tissues (e.g. VNC) could produce their own nidogen.
To ask whether nidogen is required for animal morphogenesis, Dai et al. create a series of nidogen mutant lines using CRISPR/Cas9 technology. Nidogen elimination does not have an effect on viability and overall organogenesis, but leads to reduced fertility, similar to previous observations in C. elegans and in contrast to nidogen double-knockout mice. This suggests that some functions of nidogen might be specific to vertebrates. Next, the authors look for tissue-specific defects due to nidogen loss. They find that although nidogen loss does not affect BM in most tissues, it leads to lack of integrity in the BM of the larval fat body and adult flight muscles (Fig II). In addition, laminin and collagen IV networks appear uncoupled in the fat body BM of nidogen mutants, in agreement with in vitro studies suggesting a role of nidogen as a linker between these networks. This was further confirmed by a detailed functional analysis of the different nidogen domains.
Finally, the authors investigate the dependency of nidogen incorporation into the BM on the other BM components. They find that the nidogen assembly requirements are tissue-specific: while only laminin is required for nidogen incorporation into the BM of embryonic gut, muscle and VNC tissues, both laminin and collagen IV are required for nidogen assembly in larval fat body.
Why I chose this preprint:
Nidogen is one of the four main BM components, whose role in BM formation and maintenance is not well known. This is partly due to seemingly contradictory observations from various in vitro and in vivo studies. Looking at different tissues during Drosophila development, Dai et al. show that nidogen structure, assembly and function is tissue-specific, which could explain its vitality for development of some tissues, and its dispensability for some others.
Questions for the authors:
This preprint shows that nidogen assembly and function is tissue-specific. It would be interesting to investigate why loss of nidogen affects larval fat body but not many other tissues. This preprint shows that the nidogen organisation is also tissue-specific. Could this organisational difference (e.g. continuous mesh-like structure in the fat body versus dotted pattern in visceral mesoderm) explain the effects of its loss in some tissues, but not others? It would also be interesting to investigate whether nidogen acts as a linker in tissues other than the fat body.
References:
- Pastor-Pareja, José C. & Xu, T. Shaping Cells and Organs in Drosophila by Opposing Roles of Fat Body-Secreted Collagen IV and Perlecan. Developmental Cell 21, 245-256 (2011).
- Kang, S.H. & Kramer, J.M. Nidogen Is Nonessential and Not Required for Normal Type IV Collagen Localization in Caenorhabditis elegans. Molecular Biology of the Cell 11, 3911-3923 (2000).
- Murshed, M. et al. The Absence of Nidogen 1 Does Not Affect Murine Basement Membrane Formation. Molecular and Cellular Biology 20, 7007-7012 (2000).
- Schymeinsky, J. et al. Gene Structure and Functional Analysis of the Mouse Nidogen-2 Gene: Nidogen-2 Is Not Essential for Basement Membrane Formation in Mice. Molecular and Cellular Biology 22, 6820-6830 (2002).
- Bader, B.L. et al. Compound Genetic Ablation of Nidogen 1 and 2 Causes Basement Membrane Defects and Perinatal Lethality in Mice. Molecular and Cellular Biology 25, 6846-6856 (2005).
doi: https://doi.org/10.1242/prelights.4638
Read preprintSign up to customise the site to your preferences and to receive alerts
Register hereAlso in the developmental biology category:
Chromatin priming and co-factor availability shape lineage response to the neuronal pioneer factor ASCL1 in pluripotency
Reinier Prosee, Jethro Lundie-Brown
Cardiac-immune microniches programme macrophage states in the regenerating heart
Theodora Stougiannou
Temporal degradation of PRC2 uncovers specific developmental dependencies
María Mariner-Faulí
preLists in the developmental biology category:
preLighters’ choice – Handpicked DevBio preprints
preLighters with expertise across developmental and stem cell biology have nominated a few developmental biology (and related) preprints they’re excited about and explain in a few paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Theodora Stougiannou et al. |
BSDB Spring Meeting: Molecules to Morphogenesis
The British Society for Developmental Biology (BSDB) Spring Meeting Molecules to Morphogenesis was held from 23–26 March 2026 at the University of Warwick (UK). This meeting brought together a vibrant community of researchers to discuss how molecular mechanisms are integrated across scales to drive morphogenesis, spanning diverse model systems and approaches. This preList contains preprints by presenters from the talk and poster sessions at the meeting. Please do get in touch at preLights@biologists.com if you notice any relevant preprints that we may have missed.
| List by | Ingrid Tsang |
Keystone Symposium on Stem Cell Models in Embryology 2026
The Keystone Symposium on Stem Cell Models in Embryology, 2026, was organised by Jun Wu (UT Southwestern), Jianping Fu (University of Michigan) and Miki Ebisuya (TU Dresden) and held at Asilomar Conference Grounds in California (US). The meeting discussed recent advances made in establishing stem-cell-based embryo models, what fundamental insights into developmental processes have been gleaned from them, as well as how they are beginning to be applied more widely. This prelist contains preprints by presenters at the talk and poster sessions at the conference, which our Reviews Editor in attendance spotted. Please do reach out to preLights@biologists.com if you notice any that we’ve missed.
| List by | Ingrid Tsang |
November in preprints – DevBio & Stem cell biology
preLighters with expertise across developmental and stem cell biology have nominated a few developmental and stem cell biology (and related) preprints posted in November they’re excited about and explain in a single paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Aline Grata et al. |
October in preprints – DevBio & Stem cell biology
Each month, preLighters with expertise across developmental and stem cell biology nominate a few recent developmental and stem cell biology (and related) preprints they’re excited about and explain in a single paragraph why. Short, snappy picks from working scientists — a quick way to spot fresh ideas, bold methods and papers worth reading in full. These preprints can all be found in the October preprint list published on the Node.
| List by | Deevitha Balasubramanian et al. |
October in preprints – Cell biology edition
Different preLighters, with expertise across cell biology, have worked together to create this preprint reading list for researchers with an interest in cell biology. This month, most picks fall under (1) Cell organelles and organisation, followed by (2) Mechanosignaling and mechanotransduction, (3) Cell cycle and division and (4) Cell migration
| List by | Matthew Davies et al. |
June in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: (1) Cell organelles and organisation (2) Cell signaling and mechanosensation (3) Genetics/gene expression (4) Biochemistry (5) Cytoskeleton
| List by | Barbora Knotkova et al. |
Keystone Symposium – Metabolic and Nutritional Control of Development and Cell Fate
This preList contains preprints discussed during the Metabolic and Nutritional Control of Development and Cell Fate Keystone Symposia. This conference was organized by Lydia Finley and Ralph J. DeBerardinis and held in the Wylie Center and Tupper Manor at Endicott College, Beverly, MA, United States from May 7th to 9th 2025. This meeting marked the first in-person gathering of leading researchers exploring how metabolism influences development, including processes like cell fate, tissue patterning, and organ function, through nutrient availability and metabolic regulation. By integrating modern metabolic tools with genetic and epidemiological insights across model organisms, this event highlighted key mechanisms and identified open questions to advance the emerging field of developmental metabolism.
| List by | Virginia Savy, Martin Estermann |
Biologists @ 100 conference preList
This preList aims to capture all preprints being discussed at the Biologists @100 conference in Liverpool, UK, either as part of the poster sessions or the (flash/short/full-length) talks.
| List by | Reinier Prosee, Jonathan Townson |
BSDB/GenSoc Spring Meeting 2024
A list of preprints highlighted at the British Society for Developmental Biology and Genetics Society joint Spring meeting 2024 at Warwick, UK.
| List by | Joyce Yu, Katherine Brown |
GfE/ DSDB meeting 2024
This preList highlights the preprints discussed at the 2024 joint German and Dutch developmental biology societies meeting that took place in March 2024 in Osnabrück, Germany.
| List by | Joyce Yu |
‘In preprints’ from Development 2022-2023
A list of the preprints featured in Development's 'In preprints' articles between 2022-2023
| List by | Alex Eve, Katherine Brown |
preLights peer support – preprints of interest
This is a preprint repository to organise the preprints and preLights covered through the 'preLights peer support' initiative.
| List by | preLights peer support |
The Society for Developmental Biology 82nd Annual Meeting
This preList is made up of the preprints discussed during the Society for Developmental Biology 82nd Annual Meeting that took place in Chicago in July 2023.
| List by | Joyce Yu, Katherine Brown |
CSHL 87th Symposium: Stem Cells
Preprints mentioned by speakers at the #CSHLsymp23
| List by | Alex Eve |
Journal of Cell Science meeting ‘Imaging Cell Dynamics’
This preList highlights the preprints discussed at the JCS meeting 'Imaging Cell Dynamics'. The meeting was held from 14 - 17 May 2023 in Lisbon, Portugal and was organised by Erika Holzbaur, Jennifer Lippincott-Schwartz, Rob Parton and Michael Way.
| List by | Helen Zenner |
9th International Symposium on the Biology of Vertebrate Sex Determination
This preList contains preprints discussed during the 9th International Symposium on the Biology of Vertebrate Sex Determination. This conference was held in Kona, Hawaii from April 17th to 21st 2023.
| List by | Martin Estermann |
Alumni picks – preLights 5th Birthday
This preList contains preprints that were picked and highlighted by preLights Alumni - an initiative that was set up to mark preLights 5th birthday. More entries will follow throughout February and March 2023.
| List by | Sergio Menchero et al. |
CellBio 2022 – An ASCB/EMBO Meeting
This preLists features preprints that were discussed and presented during the CellBio 2022 meeting in Washington, DC in December 2022.
| List by | Nadja Hümpfer et al. |
2nd Conference of the Visegrád Group Society for Developmental Biology
Preprints from the 2nd Conference of the Visegrád Group Society for Developmental Biology (2-5 September, 2021, Szeged, Hungary)
| List by | Nándor Lipták |
Fibroblasts
The advances in fibroblast biology preList explores the recent discoveries and preprints of the fibroblast world. Get ready to immerse yourself with this list created for fibroblasts aficionados and lovers, and beyond. Here, my goal is to include preprints of fibroblast biology, heterogeneity, fate, extracellular matrix, behavior, topography, single-cell atlases, spatial transcriptomics, and their matrix!
| List by | Osvaldo Contreras |
EMBL Synthetic Morphogenesis: From Gene Circuits to Tissue Architecture (2021)
A list of preprints mentioned at the #EESmorphoG virtual meeting in 2021.
| List by | Alex Eve |
EMBL Conference: From functional genomics to systems biology
Preprints presented at the virtual EMBL conference "from functional genomics and systems biology", 16-19 November 2020
| List by | Jesus Victorino |
Single Cell Biology 2020
A list of preprints mentioned at the Wellcome Genome Campus Single Cell Biology 2020 meeting.
| List by | Alex Eve |
Society for Developmental Biology 79th Annual Meeting
Preprints at SDB 2020
| List by | Irepan Salvador-Martinez, Martin Estermann |
FENS 2020
A collection of preprints presented during the virtual meeting of the Federation of European Neuroscience Societies (FENS) in 2020
| List by | Ana Dorrego-Rivas |
Planar Cell Polarity – PCP
This preList contains preprints about the latest findings on Planar Cell Polarity (PCP) in various model organisms at the molecular, cellular and tissue levels.
| List by | Ana Dorrego-Rivas |
Cell Polarity
Recent research from the field of cell polarity is summarized in this list of preprints. It comprises of studies focusing on various forms of cell polarity ranging from epithelial polarity, planar cell polarity to front-to-rear polarity.
| List by | Yamini Ravichandran |
TAGC 2020
Preprints recently presented at the virtual Allied Genetics Conference, April 22-26, 2020. #TAGC20
| List by | Maiko Kitaoka et al. |
3D Gastruloids
A curated list of preprints related to Gastruloids (in vitro models of early development obtained by 3D aggregation of embryonic cells). Updated until July 2021.
| List by | Paul Gerald L. Sanchez and Stefano Vianello |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
EDBC Alicante 2019
Preprints presented at the European Developmental Biology Congress (EDBC) in Alicante, October 23-26 2019.
| List by | Sergio Menchero et al. |
EMBL Seeing is Believing – Imaging the Molecular Processes of Life
Preprints discussed at the 2019 edition of Seeing is Believing, at EMBL Heidelberg from the 9th-12th October 2019
| List by | Dey Lab |
SDB 78th Annual Meeting 2019
A curation of the preprints presented at the SDB meeting in Boston, July 26-30 2019. The preList will be updated throughout the duration of the meeting.
| List by | Alex Eve |
Lung Disease and Regeneration
This preprint list compiles highlights from the field of lung biology.
| List by | Rob Hynds |
Young Embryologist Network Conference 2019
Preprints presented at the Young Embryologist Network 2019 conference, 13 May, The Francis Crick Institute, London
| List by | Alex Eve |
Pattern formation during development
The aim of this preList is to integrate results about the mechanisms that govern patterning during development, from genes implicated in the processes to theoritical models of pattern formation in nature.
| List by | Alexa Sadier |
BSCB/BSDB Annual Meeting 2019
Preprints presented at the BSCB/BSDB Annual Meeting 2019
| List by | Dey Lab |
Zebrafish immunology
A compilation of cutting-edge research that uses the zebrafish as a model system to elucidate novel immunological mechanisms in health and disease.
| List by | Shikha Nayar |











 (No Ratings Yet)
(No Ratings Yet)