Transcription repressor protein ZBTB25 interacts with HDAC1 in macrophages infected with Mycobacterium tuberculosis, and its inhibition leads to autophagy and killing of the intracellular pathogen
Posted on: 25 July 2020
Preprint posted on 22 June 2020
Article now published in mSphere at http://dx.doi.org/10.1128/msphere.00036-21
Tamoxifen reduces inflammatory infiltration of neutrophils in the airways
Posted on:
Preprint posted on 20 June 2020
Burning breath: researchers characterise the pathways relating to airway inflammation and their potential implications for respiratory disease
Selected by Zhang-He Goh, Josie GibsonCategories: immunology, microbiology, pharmacology and toxicology
preLight Authors’ note
This is the first preLight of a three-part series on airway inflammation, infection, autophagy and its mediators. Links to the second and third preLights.
Background of preprints
Tuberculosis, an infectious lung disease that affects millions of patients worldwide, is caused by the pathogen Mycobacterium tuberculosis. Unfortunately, despite the prevalent use of various combinations of drugs active against different stages of M. tuberculosis growth, tuberculosis remains an intractable infection globally.
The damage caused by the resulting immunological and inflammatory host response [1], has driven intensive research into host-directed therapies [2,3]. Targeting the host immune system affords two strategies to target tuberculosis: the first involves promoting the clearance of the infection by enhancing the immune system [4], while the second limits organ damage and immune exhaustion by managing inflammation [2].
Previous research showed that intracellular pathogens suppress host immune functions by modulating host chromatin and epigenetic regulators [5]. In fact, M. tuberculosis increases the expression of HDAC1 and decreases the expression of IL-12B, thus reducing host inflammatory responses [6]. In their preprint, Madhavan et al. further probed the mechanism by which M. tuberculosis modulates these two host proteins (Fig. 1). The authors show that manipulation of IL-12B expression by M. tuberculosis promotes pathogen survival by reducing autophagy. This is an important degradation pathway in macrophages to eliminate intracellular M. tuberculosis that we describe in our next preLight.
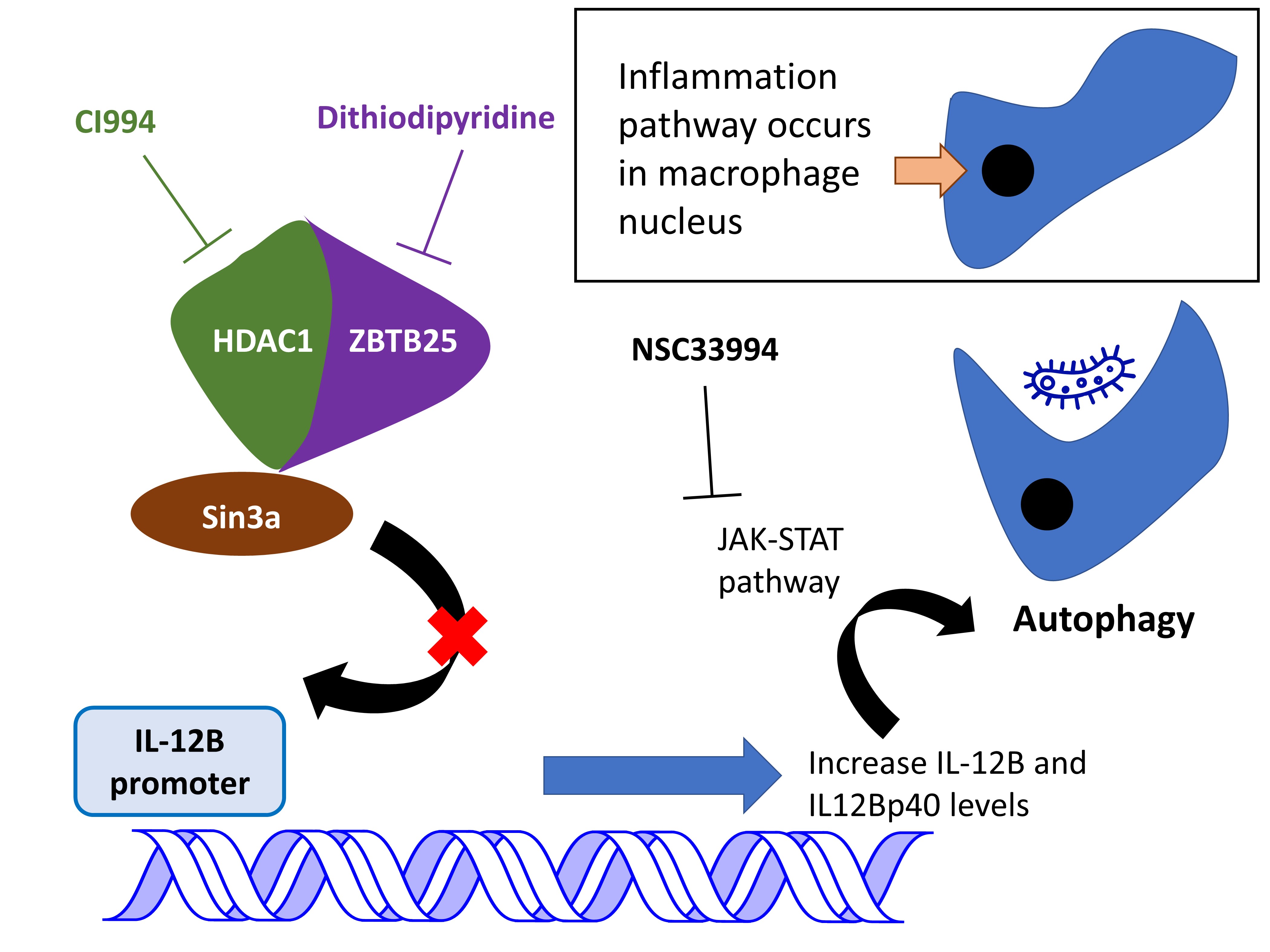
Figure 1. Mediators of autophagy in macrophages infected with M. tuberculosis described by Madhavan et al.. CI994 and dithiodipyridine induce autophagy; NSC33994 attenuates this induction.
Indeed, the role of respiratory tract inflammation extends beyond infectious diseases to respiratory diseases in general—respiratory tract inflammation caused by neutrophil infiltration has also been implicated in the aetiology of cystic fibrosis and chronic obstructive pulmonary disease. One agent that could be useful in reducing this damaging inflammation is tamoxifen [7], an anticancer drug with non-steroidal anti-inflammatory agent (NSAID) properties [8]. In their preprint, Mansilla et al. used mouse models to evaluate tamoxifen’s potential to reduce neutrophil infiltration of the airways.
Key findings of preprints
(A) Madhavan et al.
The authors found that HDAC1 in macrophages were phosphorylated upon infection by M. tuberculosis; HDAC1 interacted with two macrophage proteins, ZBTB25 and Sin3a. The interaction between HDAC1 and ZBTB25 was characterised using confocal microscopy, which showed that HDAC1 and ZBTB25 co-localise in the nucleus. In silico docking analysis also revealed a strong interaction between HDAC1 and ZBTB25. The authors then showed that ZBTB25 and Sin3a interact with the IL-12B promoter, and used a rescue experiment involving the knockdown of ZBTB25 to reinforce this observation.
Having ascertained that the modulation of ZBTB25 and HDAC1 affect the intracellular survival of M. tuberculosis, Madhavan et al. then investigated how this might be modulated by the effects of two small molecules, dithiodipyridine (ZBTB25 inhibitor) and CI994 (HDAC1 inhibitor). Indeed, the administration of dithiodipyridine and CI994 prevented the recruitment of the HDAC1, ZBTB25, and CI994 to the IL-12B promoter, disrupted the co-localisation of ZBTB25 and HDAC1 in the nucleus, and increased IL-12B expression and IL-12Bp40 levels.
The authors then linked the mechanisms of action of dithiodipyridine and CI994 to autophagy. They looked at three marker proteins involved in autophagy—ATG5, Beclin 1, and LC3—which are suppressed in macrophages infected with M. tuberculosis. Through the restoration of IL-12B expression, the administration of dithiodipyridine and CI994 restored the cells’ autophagy functions. In a separate rescue experiment involving the use of NSC33994, a specific JAK2 inhibitor, the authors showed that the JAK-STAT pathway was involved in the restoration of ATG5, Beclin 1, and LC3 expression. Together these experiments suggest inhibiting M. tuberculosis manipulation of inflammatory and autophagy pathways represent a useful host-directed therapy for tuberculosis treatment.
(B) Mansilla et al.
Mansilla et al. showed that tamoxifen reduces airway neutrophilia in acute lung injury and in chronic muco-obstructive lung disease. In both models, tamoxifen decreased neutrophilia, but did not affect macrophages and lymphocyte numbers. Because tamoxifen blocks ion channels, the authors conducted another experiment to assess its potential role in cystic fibrosis, and found that tamoxifen does not affect anion secretion in mice trachea when applied to the apical side.
Why we selected these preprints
In a previous preLight, one of us (Z.G.) wrote about a preprint by Bigelow et al. on evaluating the toxicity and efficacy of a linezolid-containing antitubercular regimen using a mouse model. In this preLight, we focus on another preprint targeting another aspect of tuberculosis: the host inflammatory responses that drive it.
There is increasing recognition that the immune system plays a key role in mediating the severity of many respiratory infections [9]; many such discoveries have most recently been made in infections caused by SARS-CoV-2 [10]. Similarly, the immune system has also been implicated in tuberculosis [2]. The preprint by Madhavan et al. serves to inform a host-directed strategy in mitigating the effects of tuberculosis infections; indeed, increasing evidence implicates macrophages in the pathology of tuberculosis [11,12]. Madhavan et al. shed some light on the mechanisms involved in these infections. Coupled with the use of other in vitro models [13,14], these findings will guide efforts that aim to attenuate potentially destructive immunological manipulation in tuberculosis.
In keeping with the theme of inflammation, we also selected the preprint by Mansilla et al., in which the authors investigated tamoxifen’s anti-inflammatory effects in the respiratory tract. Their findings that tamoxifen appears to affect only the infiltration of neutrophils, but not the infiltration of macrophages nor lymphocytes, could serve as starting points by which researchers design anti-inflammatory agents to manage respiratory diseases.
Future work
Following the characterisation of the in vitro mechanisms underlying inflammation and autophagy in tuberculosis by Madhavan et al., the next step would be to test these hypotheses in vivo. Animal models, which are generally more complex than in vitro systems, will be needed to confirm the pathways through which inflammation and autophagy arise.
Yet, as described in the preprint by Mansilla et al., the immune response to anti-inflammatory agents in vivo is complex. This gives rise to a second issue—the clinical targetability of the immune system in infectious diseases like tuberculosis. Host-directed therapies will need to achieve a fine balance between attenuating the damaging immune response in the lung, and preserving the protective nature of the body’s immune response. Safety, as always, must remain a top priority.
Questions for authors
(A) Madhavan et al.
- You showed that HDAC1 co-localises with ZBTB25 (preprint Figure 1D), which led to further in silico docking experiments. Were any co-localisation or in silico experiments also conducted with Sin3a? Why did you choose to focus on ZBTB25 rather than Sin3a?
- How do dithiodipyridine and CI994 bind to ZBTB25 and HDAC1 respectively, and how do they disrupt the protein-protein interactions between them? How might they inform future efforts to develop clinically relevant inhibitors for these targets?
- The role of autophagy in clearance of tuberculosis was focussed on macrophages. Do you know if similar mechanisms may be important for other infected cell types?
(B) Mansilla et al.
- Interestingly, tamoxifen has also been reported to modulate the immune system both in humans and in mouse models [15], with differential effects on different immune cell subtypes. In your experiments, did the effects of tamoxifen on cell infiltration also vary across different lymphoctyes (e.g. B cells, helper T cells, etc.)?
References
[1] Ndlovu H, Marakalala MJ, Granulomas and Inflammation: Host-Directed Therapies for Tuberculosis, Frontiers in Immunology 7(434) (2016).
[2] Zumla A, Rao M, Parida SK, Keshavjee S, Cassell G, Wallis R, Axelsson-Robertsson R, Doherty M, Andersson J, Maeurer M, Inflammation and tuberculosis: host-directed therapies, Journal of Internal Medicine 277(4) (2015) 373-387.
[3] Kaufmann SHE, Dorhoi A, Hotchkiss RS, Bartenschlager R, Host-directed therapies for bacterial and viral infections, Nat Rev Drug Discov 17(1) (2018) 35-56.
[4] Young C, Walzl G, Du Plessis N, Therapeutic host-directed strategies to improve outcome in tuberculosis, Mucosal Immunology 13(2) (2020) 190-204.
[5] Kathirvel M, Mahadevan S, The role of epigenetics in tuberculosis infection, Epigenomics 8(4) (2016) 537-549.
[6] Chandran A, Antony C, Jose L, Mundayoor S, Natarajan K, Kumar RA, Mycobacterium tuberculosis Infection Induces HDAC1-Mediated Suppression of IL-12B Gene Expression in Macrophages, Frontiers in Cellular and Infection Microbiology 5(90) (2015).
[7] Perez B, Henriquez C, Sarmiento J, Morales N, Folch H, Galesio JS, Uberti B, Morán G, Tamoxifen as a new therapeutic tool for neutrophilic lung inflammation, Respirology 21(1) (2016) 112-118.
[8] Takada Y, Bhardwaj A, Potdar P, Aggarwal BB, Nonsteroidal anti-inflammatory agents differ in their ability to suppress NF-κB activation, inhibition of expression of cyclooxygenase-2 and cyclin D1, and abrogation of tumor cell proliferation, Oncogene 23(57) (2004) 9247-9258.
[9] Chen K, Kolls JK, T cell-mediated host immune defenses in the lung, Annu Rev Immunol 31 (2013) 605-633.
[10] Polidoro RB, Hagan RS, de Santis Santiago R, Schmidt NW, Overview: Systemic Inflammatory Response Derived From Lung Injury Caused by SARS-CoV-2 Infection Explains Severe Outcomes in COVID-19, Frontiers in Immunology 11(1626) (2020).
[11] Khan A, Singh VK, Hunter RL, Jagannath C, Macrophage heterogeneity and plasticity in tuberculosis, Journal of Leukocyte Biology 106(2) (2019) 275-282.
[12] Guirado E, Schlesinger LS, Kaplan G, Macrophages in tuberculosis: friend or foe, Semin Immunopathol 35(5) (2013) 563-583.
[13] Kapoor N, Pawar S, Sirakova TD, Deb C, Warren WL, Kolattukudy PE, Human Granuloma In Vitro Model, for TB Dormancy and Resuscitation, PLOS ONE 8(1) (2013) e53657.
[14] Crouser ED, White P, Caceres EG, Julian MW, Papp AC, Locke LW, Sadee W, Schlesinger LS, A Novel In Vitro Human Granuloma Model of Sarcoidosis and Latent Tuberculosis Infection, Am J Respir Cell Mol Biol 57(4) (2017) 487-498.
[15] Behjati S, Frank MH, The effects of tamoxifen on immunity, Curr Med Chem 16(24) (2009) 3076-3080.
Sign up to customise the site to your preferences and to receive alerts
Register hereAlso in the immunology category:
Gut microbiome changes over the course of multiple sclerosis differentially influence autoimmune neuroinflammation
Carole Djagang et al.
Human Assembloid Model of Emergent Neurotropic Enteroviruses
Mitchell Sarmie
Small Molecule Agonists of TREM2 Reprogram Microglia and Protect Synapses in Human Alzheimer’s Models
Dina Kabbara
Also in the microbiology category:
Gut microbiome changes over the course of multiple sclerosis differentially influence autoimmune neuroinflammation
Carole Djagang et al.
Human Assembloid Model of Emergent Neurotropic Enteroviruses
Mitchell Sarmie
Cryo-EM reveals multiple mechanisms of ribosome inhibition by doxycycline
Leonie Brüne
Also in the pharmacology and toxicology category:
Small Molecule Agonists of TREM2 Reprogram Microglia and Protect Synapses in Human Alzheimer’s Models
Dina Kabbara
Snake venom metalloproteinases are predominantly responsible for the cytotoxic effects of certain African viper venoms
Daniel Osorno Valencia
In vitro pharmacokinetics and pharmacodynamics of the diarylquinoline TBAJ-587 and its metabolites against Mycobacterium tuberculosis
Zhang-He Goh
preLists in the immunology category:
BSDB Spring Meeting: Molecules to Morphogenesis
The British Society for Developmental Biology (BSDB) Spring Meeting Molecules to Morphogenesis was held from 23–26 March 2026 at the University of Warwick (UK). This meeting brought together a vibrant community of researchers to discuss how molecular mechanisms are integrated across scales to drive morphogenesis, spanning diverse model systems and approaches. This preList contains preprints by presenters from the talk and poster sessions at the meeting. Please do get in touch at preLights@biologists.com if you notice any relevant preprints that we may have missed.
| List by | Ingrid Tsang |
SciELO preprints – From 2025 onwards
SciELO has become a cornerstone of open, multilingual scholarly communication across Latin America. Its preprint server, SciELO preprints, is expanding the global reach of preprinted research from the region (for more information, see our interview with Carolina Tanigushi). This preList brings together biological, English language SciELO preprints to help readers discover emerging work from the Global South. By highlighting these preprints in one place, we aim to support visibility, encourage early feedback, and showcase the vibrant research communities contributing to SciELO’s open science ecosystem.
| List by | Carolina Tanigushi |
Community-driven preList – Immunology
In this community-driven preList, a group of preLighters, with expertise in different areas of immunology have worked together to create this preprint reading list.
| List by | Felipe Del Valle Batalla et al. |
Journal of Cell Science meeting ‘Imaging Cell Dynamics’
This preList highlights the preprints discussed at the JCS meeting 'Imaging Cell Dynamics'. The meeting was held from 14 - 17 May 2023 in Lisbon, Portugal and was organised by Erika Holzbaur, Jennifer Lippincott-Schwartz, Rob Parton and Michael Way.
| List by | Helen Zenner |
Fibroblasts
The advances in fibroblast biology preList explores the recent discoveries and preprints of the fibroblast world. Get ready to immerse yourself with this list created for fibroblasts aficionados and lovers, and beyond. Here, my goal is to include preprints of fibroblast biology, heterogeneity, fate, extracellular matrix, behavior, topography, single-cell atlases, spatial transcriptomics, and their matrix!
| List by | Osvaldo Contreras |
Single Cell Biology 2020
A list of preprints mentioned at the Wellcome Genome Campus Single Cell Biology 2020 meeting.
| List by | Alex Eve |
Autophagy
Preprints on autophagy and lysosomal degradation and its role in neurodegeneration and disease. Includes molecular mechanisms, upstream signalling and regulation as well as studies on pharmaceutical interventions to upregulate the process.
| List by | Sandra Malmgren Hill |
Antimicrobials: Discovery, clinical use, and development of resistance
Preprints that describe the discovery of new antimicrobials and any improvements made regarding their clinical use. Includes preprints that detail the factors affecting antimicrobial selection and the development of antimicrobial resistance.
| List by | Zhang-He Goh |
Zebrafish immunology
A compilation of cutting-edge research that uses the zebrafish as a model system to elucidate novel immunological mechanisms in health and disease.
| List by | Shikha Nayar |
Also in the microbiology category:
SciELO preprints – From 2025 onwards
SciELO has become a cornerstone of open, multilingual scholarly communication across Latin America. Its preprint server, SciELO preprints, is expanding the global reach of preprinted research from the region (for more information, see our interview with Carolina Tanigushi). This preList brings together biological, English language SciELO preprints to help readers discover emerging work from the Global South. By highlighting these preprints in one place, we aim to support visibility, encourage early feedback, and showcase the vibrant research communities contributing to SciELO’s open science ecosystem.
| List by | Carolina Tanigushi |
BioMalPar XVI: Biology and Pathology of the Malaria Parasite
[under construction] Preprints presented at the (fully virtual) EMBL BioMalPar XVI, 17-18 May 2020 #emblmalaria
| List by | Dey Lab, Samantha Seah |
1
ECFG15 – Fungal biology
Preprints presented at 15th European Conference on Fungal Genetics 17-20 February 2020 Rome
| List by | Hiral Shah |
EMBL Seeing is Believing – Imaging the Molecular Processes of Life
Preprints discussed at the 2019 edition of Seeing is Believing, at EMBL Heidelberg from the 9th-12th October 2019
| List by | Dey Lab |
Antimicrobials: Discovery, clinical use, and development of resistance
Preprints that describe the discovery of new antimicrobials and any improvements made regarding their clinical use. Includes preprints that detail the factors affecting antimicrobial selection and the development of antimicrobial resistance.
| List by | Zhang-He Goh |
Also in the pharmacology and toxicology category:
SciELO preprints – From 2025 onwards
SciELO has become a cornerstone of open, multilingual scholarly communication across Latin America. Its preprint server, SciELO preprints, is expanding the global reach of preprinted research from the region (for more information, see our interview with Carolina Tanigushi). This preList brings together biological, English language SciELO preprints to help readers discover emerging work from the Global South. By highlighting these preprints in one place, we aim to support visibility, encourage early feedback, and showcase the vibrant research communities contributing to SciELO’s open science ecosystem.
| List by | Carolina Tanigushi |
April in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) biochemistry/metabolism 2) cell cycle and division 3) cell organelles and organisation 4) cell signalling and mechanosensing 5) (epi)genetics
| List by | Vibha SINGH et al. |
FENS 2020
A collection of preprints presented during the virtual meeting of the Federation of European Neuroscience Societies (FENS) in 2020
| List by | Ana Dorrego-Rivas |
COVID-19 / SARS-CoV-2 preprints
List of important preprints dealing with the ongoing coronavirus outbreak. See http://covidpreprints.com for additional resources and timeline, and https://connect.biorxiv.org/relate/content/181 for full list of bioRxiv and medRxiv preprints on this topic
| List by | Dey Lab, Zhang-He Goh |
1
Drug use in special populations
Any drugs that are being used in special populations: Patients with liver and kidney failure, in paediatrics, in geriatrics, and in pregnant or lactating patients. Includes the discovery of factors that could potentially affect drug use in these special populations.
| List by | Zhang-He Goh |
Toxicology of toxicants, existing therapeutics, and investigational drugs
Preprints that describe the toxicology of environmental pollutants and existing and upcoming drugs. Includes both toxicokinetics and toxicodynamics, as well as technological improvements that will help in the characterisation of this field.
| List by | Zhang-He Goh |
Anticancer agents: Discovery and clinical use
Preprints that describe the discovery of anticancer agents and their clinical use. Includes both small molecules and macromolecules like biologics.
| List by | Zhang-He Goh |
Advances in Drug Delivery
Advances in formulation technology or targeted delivery methods that describe or develop the distribution of small molecules or large macromolecules to specific parts of the body.
| List by | Zhang-He Goh |











 (No Ratings Yet)
(No Ratings Yet)