Detergent-Triggered Membrane Remodelling Monitored via Intramembrane Fluorescence De-Quenching
Posted on: 25 March 2026
Preprint posted on 26 September 2025
Article now published in at https://pubs.acs.org/doi/10.1021/acsomega.5c10435
A sensitive and accessible method to probe detergent–membrane interactions that also advances the study of lipid–protein dynamics
Selected by Cyntia Alves Conceição, Marcus OliveiraCategories: bioengineering, biophysics, molecular biology
Summary/Background:
The composition and functionality of biological membranes are of great importance for cell maintenance. Additionally, mimetic membranes are widely used for production of drugs and vaccines, and in in vitro studies with different purposes1–3.
Integral membrane proteins (IMPs) are crucial for life, as they perform vital functions like transporting molecules, transmitting signals, and facilitating communication across cell membranes4. To study these IMPs, it is crucial to purify them from cellular membranes without compromising their functions. Moreover, membrane solubilization is important for biotechnological applications such as virus inactivation, cellular drug delivery and formation of niosomes in vaccine formulations5–7.
Triton X-100 (TX-100) is a non-ionic detergent used for IMP isolation, while maintaining their structure and function8. However, the mechanisms of detergent-membrane solubilization are still poorly understood. Several aspects of their interaction, which depends on membrane fusion9, are still unclear — including how exactly the detergent acts at the molecular level, how it affects lipid–lipid interactions, and what the structure and morphology of the fused products are.
In recent years, a three-step model has been proposed: (1) detergent monomers, at concentrations below the critical micellar concentration (CMC), progressively saturate the membrane; (2) the formation of mixed detergent–lipid micelles lead to membrane disruption and fragmentation; and (3) these mixed micelles are subsequently released into the solution9. This preprint builds on the author’s previous study using Förster resonance energy transfer (FRET) and single-vesicle analysis to investigate how Triton X-100 (TX-100) interacts with synthetic large unilamellar vesicles (LUVs)10. In this preprint, the authors present a fluorescence de-quenching assay to confirm the presence of a vesicle swelling step that occurs prior to complete solubilization or micellization.
Significance/Why I think this preprint is important:
This study is highly significant, not only for elucidating how detergents interact with and solubilize membranes, but also for providing the scientific community with a robust technique to assess and characterize lipid–protein interactions. In the context of detergent–membrane solubilization, the approach presented in this preprint is both sensitive and relatively straightforward to apply and interpret, particularly when compared to methods such as FRET analysis. Collectively, these aspects underscore the study’s relevance for advancing our understanding of biological membrane behavior.
Key findings:
Fluorescence de-quenching tracks TX-100–induced membrane perturbations
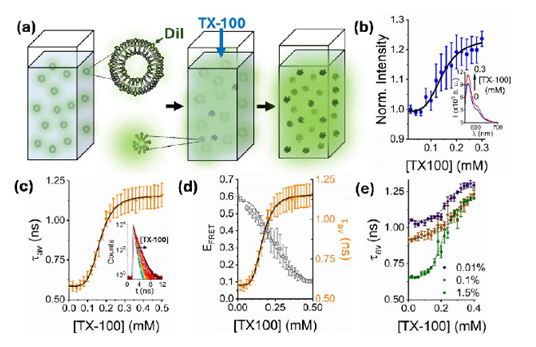
In preprint Figure 1, the authors show that increasing concentrations of TX-100 led to an increase in DiI fluorescence emission, along with a rise in the average lifetime amplitude (τₐᵥ), suggesting that DiI fluorescence de-quenching occurs proportionally to the amount of TX-100 added to POPC membranes. The authors further compared the observed de-quenching effects with FRET data from their previous study, in which the FRET signal was shown to decrease following detergent–membrane interaction. Collectively, these results support the reliability of the de-quenching approach as a quantitative method for detecting nanoscale perturbations in vesicles.
Extending fluorescence dequenching to other detergent-induced membrane disturbances
With the goal of extending the applicability of their technique to experiments using other fluorescent probes, the authors used DiO and DiD dyes (shown in preprint Figure 2), which also had their emission intensities and ͳav increased. Variations in the magnitude of the lifetime changes across the dyes were correlated by the authors with potential differences in dye–lipid binding strength, intermolecular dye interactions within the membrane, and the extent of dye incorporation into the bilayer. Furthermore, the authors used other surfactants, such as sodium dodecyl sulfate (SDS) and Tween 20 to assess the utility of de-quenching measurements across a range of disruptors. With this data, the authors show that fluorescence de-quenching may be a generalizable assay for assessing detergent-membrane solubilization.
Vesicle imaging corroborates dequenching findings
The authors used fluorescence lifetime imaging (FLIM) to visualize vesicles de-quenching and assess lifetime distributions in Dil-labelled LUVs (preprint Figure 3). The authors observed a clear difference in the fluorophore decay time in the presence of the detergent, corroborating the DiI emission de-quenching and alterations in the lipid microenvironment, as previously shown in preprint Figure 1. As illustrated in preprint Figure S3, the total vesicle count remained stable, suggesting that the fluorescence variations result from morphological rearrangements rather than vesicle loss. To test this hypothesis, the authors employed scanning electron microscopy to visualize LUVs in the absence and presence of TX-100 (preprint Figure 4). The images revealed that exposure to TX-100 increased vesicle diameter and induced a morphological transition from spherical to toroidal-like structures. Taken together, the findings support the previously proposed solubilization model, in which TX-100 promotes vesicle swelling and morphological rearrangements preceding the full micellization and solubilization of LUV membranes.
Comments/Questions:
Major comments
- The manuscript would benefit from the inclusion of a statistical analysis section within the Methods, specifying the tests used and the criteria for data significance. This addition would enhance the transparency and reproducibility of the results.
- In the first paragraph of the results the authors mentioned that the distance between the probes inserted in POPC vesicles’ bilayers (DiI–DiI) was <2 nm. However, it is not clear how this distance was determined. Is there an optimal or required distance between the probes to assess its quenching mode? And it is not clear whether the distances were also determined for the other probes used (DiD and DiO), and whether these values were comparable to those observed for DiI–DiI. Including this information would enhance the reproducibility and demonstrate the broader applicability of the assay.
- In preprint Figures 1 and 2, increasing concentrations of TX-100 were used. How do the authors ensure that concentrations below the CMC do not disrupt the membranes prior to micellization? It might be helpful to include vesicle morphology data at detergent concentrations below the CMC, obtained by scanning electron microscopy, to clarify this point. Moreover, it is not clear how the detergent concentrations used in preprint Figures 3 and 4 were selected, and how these differences could influence the interpretation of the results. Providing data or representative images that demonstrate the micellization process under the experimental conditions used in this study would further strengthen the work.
- The authors could consider replacing the representative microscopy images in preprint Figure 3 with higher-resolution versions, particularly to improve the visibility of the scale bar.
- Greater emphasis on the statistical analysis of preprint Figures 3 and 4 is recommended. While the authors report the number of vesicles analyzed in the graphs, it remains unclear whether the results were reproducible across independent vesicle preparations. Including such information would enhance the credibility and interpretability of the data.
- The manuscript would benefit from a discussion on how its results align or contrast with other studies utilizing quenching techniques to assess LUV structure and surfactant interactions (g.: PMIDs: 36901959; 39132807; 8580339; 34150932). Such comparisons would provide important context for interpreting the findings.
Minor comments
- Replace “remodelling” with “remodeling.”
- Including page numbers throughout the document is recommended.
- The experiments and figures in the preprint align well with the authors’ aims. However, improving the resolution of the figures would enhance the overall impact of the data presentation.
- In preprint Figure 1e, the authors present the optimization of the probe concentration used in the study. The figure shows three concentrations (0.01%, 0.1%, and 1.5%), none of which correspond to the concentration used in the experiments, which may be confusing for readers. It would be helpful to include the curve corresponding to the concentration employed in most of the experiments (1%).
- The meaning of the red and black lines in preprint Figure 1e is not clearly explained. Clarifying what these lines represent would help readers better interpret the data presented.
- The use of different fluorescent probes supports the notion that the de-quenching assay can be generalized beyond DiI/TX-100, as shown in preprint Figure 2. However, the manuscript does not specify the sources of these probes or describe how they differ structurally or in their interactions with membranes. Including this information would provide stronger support for the discussion, as such differences could explain the variations in fluorescence emissions and quantum yield observed in Figure 2e.
- Although the overall results support the idea that vesicle swelling occurs as an intermediate step before micellization, the use of terms such as “indicating” (page 7, 2nd paragraph) and “assigned” (page 9) may give the impression that this phenomenon was directly observed in all figures, whereas it becomes visually evident only toward the end of the manuscript. Revising the text or adjusting the order of the figures could help highlight this key finding more clearly for the reader.
- It appears that some references are missing in sections where the authors cite previous findings, particularly in the Discussion (page 11, 3rd paragraph). Including these and other citations would provide proper context and strengthen the manuscript.
- Including a graphical abstract illustrating the proposed vesicle swelling step could greatly aid readers in visualizing and understanding the central mechanistic concept of the study.
References:
- Chugh, V., Vijaya Krishna, K. & Pandit, A. Cell Membrane-Coated Mimics: A Methodological Approach for Fabrication, Characterization for Therapeutic Applications, and Challenges for Clinical Translation. ACS Nano 15, 17080–17123 (2021).
- Majeed, S., Ahmad, A. B., Sehar, U. & Georgieva, E. R. Lipid Membrane Mimetics in Functional and Structural Studies of Integral Membrane Proteins. Membranes (Basel). 11, (2021).
- Tan, S., Wu, T., Zhang, D. & Zhang, Z. Cell or cell membrane-based drug delivery systems. Theranostics 5, 863–81 (2015).
- Whitelegge, J. P. Integral membrane proteins and bilayer proteomics. Anal. Chem. 85, 2558–68 (2013).
- Lin, Q. et al. Sanitizing agents for virus inactivation and disinfection. View (Beijing, China) 1, e16 (2020).
- Hammond, J. et al. Membrane Fusion-Based Drug Delivery Liposomes Transiently Modify the Material Properties of Synthetic and Biological Membranes. Small 21, e2408039 (2025).
- Liga, S., Paul, C., Moacă, E.-A. & Péter, F. Niosomes: Composition, Formulation Techniques, and Recent Progress as Delivery Systems in Cancer Therapy. Pharmaceutics 16, (2024).
- Koley, D. & Bard, A. J. Triton X-100 concentration effects on membrane permeability of a single HeLa cell by scanning electrochemical microscopy (SECM). Proc. Natl. Acad. Sci. U. S. A. 107, 16783–7 (2010).
- Lichtenberg, D., Ahyayauch, H. & Goñi, F. M. The mechanism of detergent solubilization of lipid bilayers. Biophys. J. 105, 289–99 (2013).
- Dresser, L. G. et al. Multiple intermediates in the detergent-induced fusion of lipid vesicles. Commun. Mater. 5, 195 (2024).
Language refinement was supported using ChatGPT (OpenAI), which assisted in improving clarity and readability.
doi: https://doi.org/10.1242/prelights.43145
Read preprintSign up to customise the site to your preferences and to receive alerts
Register hereAlso in the bioengineering category:
A Novel Chimeric Antigen Receptor (CAR) - Strategy to Target EGFRVIII-Mutated Glioblastoma Cells via Macrophages
Dina Kabbara
Human pluripotent stem cell-derived macrophages modify development of human kidney organoids
Theodora Stougiannou
Matrix viscoelasticity regulates dendritic cell migration and immune priming
Roberto Amadio
Also in the biophysics category:
Mechanically-induced Septin Networks Protect Nuclear Integrity
Filipe Nunes Vicente
Loss of Sun2 ablates nuclear mechanosensing-driven extracellular matrix production and mitigates lung fibrosis
Beth Chopak
Shape independent fluidisation in epithelial monolayers
Sindhu Muthukrishnan
Also in the molecular biology category:
Chromatin priming and co-factor availability shape lineage response to the neuronal pioneer factor ASCL1 in pluripotency
Reinier Prosee, Jethro Lundie-Brown
Temporal degradation of PRC2 uncovers specific developmental dependencies
María Mariner-Faulí
Remote homology and functional genetics unmask deeply preserved Scm3/HJURP orthologs in metazoans
Reinier Prosee
preLists in the bioengineering category:
October in preprints – DevBio & Stem cell biology
Each month, preLighters with expertise across developmental and stem cell biology nominate a few recent developmental and stem cell biology (and related) preprints they’re excited about and explain in a single paragraph why. Short, snappy picks from working scientists — a quick way to spot fresh ideas, bold methods and papers worth reading in full. These preprints can all be found in the October preprint list published on the Node.
| List by | Deevitha Balasubramanian et al. |
CSHL 87th Symposium: Stem Cells
Preprints mentioned by speakers at the #CSHLsymp23
| List by | Alex Eve |
EMBL Synthetic Morphogenesis: From Gene Circuits to Tissue Architecture (2021)
A list of preprints mentioned at the #EESmorphoG virtual meeting in 2021.
| List by | Alex Eve |
3D Gastruloids
A curated list of preprints related to Gastruloids (in vitro models of early development obtained by 3D aggregation of embryonic cells). Updated until July 2021.
| List by | Paul Gerald L. Sanchez and Stefano Vianello |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
EMBL Seeing is Believing – Imaging the Molecular Processes of Life
Preprints discussed at the 2019 edition of Seeing is Believing, at EMBL Heidelberg from the 9th-12th October 2019
| List by | Dey Lab |
Lung Disease and Regeneration
This preprint list compiles highlights from the field of lung biology.
| List by | Rob Hynds |
Advances in microscopy
This preList highlights exciting unpublished preprint articles describing advances in microscopy with a focus on light-sheet microscopy.
| List by | Stephan Daetwyler |
Also in the biophysics category:
October in preprints – DevBio & Stem cell biology
Each month, preLighters with expertise across developmental and stem cell biology nominate a few recent developmental and stem cell biology (and related) preprints they’re excited about and explain in a single paragraph why. Short, snappy picks from working scientists — a quick way to spot fresh ideas, bold methods and papers worth reading in full. These preprints can all be found in the October preprint list published on the Node.
| List by | Deevitha Balasubramanian et al. |
October in preprints – Cell biology edition
Different preLighters, with expertise across cell biology, have worked together to create this preprint reading list for researchers with an interest in cell biology. This month, most picks fall under (1) Cell organelles and organisation, followed by (2) Mechanosignaling and mechanotransduction, (3) Cell cycle and division and (4) Cell migration
| List by | Matthew Davies et al. |
March in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) cancer biology 2) cell migration 3) cell organelles and organisation 4) cell signalling and mechanosensing 5) genetics and genomics 6) other
| List by | Girish Kale et al. |
Biologists @ 100 conference preList
This preList aims to capture all preprints being discussed at the Biologists @100 conference in Liverpool, UK, either as part of the poster sessions or the (flash/short/full-length) talks.
| List by | Reinier Prosee, Jonathan Townson |
February in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) biochemistry and cell metabolism 2) cell organelles and organisation 3) cell signalling, migration and mechanosensing
| List by | Barbora Knotkova et al. |
preLights peer support – preprints of interest
This is a preprint repository to organise the preprints and preLights covered through the 'preLights peer support' initiative.
| List by | preLights peer support |
66th Biophysical Society Annual Meeting, 2022
Preprints presented at the 66th BPS Annual Meeting, Feb 19 - 23, 2022 (The below list is not exhaustive and the preprints are listed in no particular order.)
| List by | Soni Mohapatra |
EMBL Synthetic Morphogenesis: From Gene Circuits to Tissue Architecture (2021)
A list of preprints mentioned at the #EESmorphoG virtual meeting in 2021.
| List by | Alex Eve |
Biophysical Society Meeting 2020
Some preprints presented at the Biophysical Society Meeting 2020 in San Diego, USA.
| List by | Tessa Sinnige |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
EMBL Seeing is Believing – Imaging the Molecular Processes of Life
Preprints discussed at the 2019 edition of Seeing is Believing, at EMBL Heidelberg from the 9th-12th October 2019
| List by | Dey Lab |
Biomolecular NMR
Preprints related to the application and development of biomolecular NMR spectroscopy
| List by | Reid Alderson |
Biophysical Society Annual Meeting 2019
Few of the preprints that were discussed in the recent BPS annual meeting at Baltimore, USA
| List by | Joseph Jose Thottacherry |
Also in the molecular biology category:
preLighters’ choice – Handpicked DevBio preprints
preLighters with expertise across developmental and stem cell biology have nominated a few developmental biology (and related) preprints they’re excited about and explain in a few paragraph why. Concise preprint highlights, prepared by the preLighter community – a quick way to spot upcoming trends, new methods and fresh ideas.
| List by | Theodora Stougiannou et al. |
BSDB Spring Meeting: Molecules to Morphogenesis
The British Society for Developmental Biology (BSDB) Spring Meeting Molecules to Morphogenesis was held from 23–26 March 2026 at the University of Warwick (UK). This meeting brought together a vibrant community of researchers to discuss how molecular mechanisms are integrated across scales to drive morphogenesis, spanning diverse model systems and approaches. This preList contains preprints by presenters from the talk and poster sessions at the meeting. Please do get in touch at preLights@biologists.com if you notice any relevant preprints that we may have missed.
| List by | Ingrid Tsang |
Keystone Symposium on Stem Cell Models in Embryology 2026
The Keystone Symposium on Stem Cell Models in Embryology, 2026, was organised by Jun Wu (UT Southwestern), Jianping Fu (University of Michigan) and Miki Ebisuya (TU Dresden) and held at Asilomar Conference Grounds in California (US). The meeting discussed recent advances made in establishing stem-cell-based embryo models, what fundamental insights into developmental processes have been gleaned from them, as well as how they are beginning to be applied more widely. This prelist contains preprints by presenters at the talk and poster sessions at the conference, which our Reviews Editor in attendance spotted. Please do reach out to preLights@biologists.com if you notice any that we’ve missed.
| List by | Ingrid Tsang |
SciELO preprints – From 2025 onwards
SciELO has become a cornerstone of open, multilingual scholarly communication across Latin America. Its preprint server, SciELO preprints, is expanding the global reach of preprinted research from the region (for more information, see our interview with Carolina Tanigushi). This preList brings together biological, English language SciELO preprints to help readers discover emerging work from the Global South. By highlighting these preprints in one place, we aim to support visibility, encourage early feedback, and showcase the vibrant research communities contributing to SciELO’s open science ecosystem.
| List by | Carolina Tanigushi |
October in preprints – DevBio & Stem cell biology
Each month, preLighters with expertise across developmental and stem cell biology nominate a few recent developmental and stem cell biology (and related) preprints they’re excited about and explain in a single paragraph why. Short, snappy picks from working scientists — a quick way to spot fresh ideas, bold methods and papers worth reading in full. These preprints can all be found in the October preprint list published on the Node.
| List by | Deevitha Balasubramanian et al. |
October in preprints – Cell biology edition
Different preLighters, with expertise across cell biology, have worked together to create this preprint reading list for researchers with an interest in cell biology. This month, most picks fall under (1) Cell organelles and organisation, followed by (2) Mechanosignaling and mechanotransduction, (3) Cell cycle and division and (4) Cell migration
| List by | Matthew Davies et al. |
September in preprints – Cell biology edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading list. This month, categories include: (1) Cell organelles and organisation, (2) Cell signalling and mechanosensing, (3) Cell metabolism, (4) Cell cycle and division, (5) Cell migration
| List by | Sristilekha Nath et al. |
June in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: (1) Cell organelles and organisation (2) Cell signaling and mechanosensation (3) Genetics/gene expression (4) Biochemistry (5) Cytoskeleton
| List by | Barbora Knotkova et al. |
May in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) Biochemistry/metabolism 2) Cancer cell Biology 3) Cell adhesion, migration and cytoskeleton 4) Cell organelles and organisation 5) Cell signalling and 6) Genetics
| List by | Barbora Knotkova et al. |
Keystone Symposium – Metabolic and Nutritional Control of Development and Cell Fate
This preList contains preprints discussed during the Metabolic and Nutritional Control of Development and Cell Fate Keystone Symposia. This conference was organized by Lydia Finley and Ralph J. DeBerardinis and held in the Wylie Center and Tupper Manor at Endicott College, Beverly, MA, United States from May 7th to 9th 2025. This meeting marked the first in-person gathering of leading researchers exploring how metabolism influences development, including processes like cell fate, tissue patterning, and organ function, through nutrient availability and metabolic regulation. By integrating modern metabolic tools with genetic and epidemiological insights across model organisms, this event highlighted key mechanisms and identified open questions to advance the emerging field of developmental metabolism.
| List by | Virginia Savy, Martin Estermann |
April in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) biochemistry/metabolism 2) cell cycle and division 3) cell organelles and organisation 4) cell signalling and mechanosensing 5) (epi)genetics
| List by | Vibha SINGH et al. |
Biologists @ 100 conference preList
This preList aims to capture all preprints being discussed at the Biologists @100 conference in Liverpool, UK, either as part of the poster sessions or the (flash/short/full-length) talks.
| List by | Reinier Prosee, Jonathan Townson |
February in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) biochemistry and cell metabolism 2) cell organelles and organisation 3) cell signalling, migration and mechanosensing
| List by | Barbora Knotkova et al. |
Community-driven preList – Immunology
In this community-driven preList, a group of preLighters, with expertise in different areas of immunology have worked together to create this preprint reading list.
| List by | Felipe Del Valle Batalla et al. |
January in preprints – the CellBio edition
A group of preLighters, with expertise in different areas of cell biology, have worked together to create this preprint reading lists for researchers with an interest in cell biology. This month, categories include: 1) biochemistry/metabolism 2) cell migration 3) cell organelles and organisation 4) cell signalling and mechanosensing 5) genetics/gene expression
| List by | Barbora Knotkova et al. |
2024 Hypothalamus GRC
This 2024 Hypothalamus GRC (Gordon Research Conference) preList offers an overview of cutting-edge research focused on the hypothalamus, a critical brain region involved in regulating homeostasis, behavior, and neuroendocrine functions. The studies included cover a range of topics, including neural circuits, molecular mechanisms, and the role of the hypothalamus in health and disease. This collection highlights some of the latest advances in understanding hypothalamic function, with potential implications for treating disorders such as obesity, stress, and metabolic diseases.
| List by | Nathalie Krauth |
BSCB-Biochemical Society 2024 Cell Migration meeting
This preList features preprints that were discussed and presented during the BSCB-Biochemical Society 2024 Cell Migration meeting in Birmingham, UK in April 2024. Kindly put together by Sara Morais da Silva, Reviews Editor at Journal of Cell Science.
| List by | Reinier Prosee |
‘In preprints’ from Development 2022-2023
A list of the preprints featured in Development's 'In preprints' articles between 2022-2023
| List by | Alex Eve, Katherine Brown |
CSHL 87th Symposium: Stem Cells
Preprints mentioned by speakers at the #CSHLsymp23
| List by | Alex Eve |
9th International Symposium on the Biology of Vertebrate Sex Determination
This preList contains preprints discussed during the 9th International Symposium on the Biology of Vertebrate Sex Determination. This conference was held in Kona, Hawaii from April 17th to 21st 2023.
| List by | Martin Estermann |
Alumni picks – preLights 5th Birthday
This preList contains preprints that were picked and highlighted by preLights Alumni - an initiative that was set up to mark preLights 5th birthday. More entries will follow throughout February and March 2023.
| List by | Sergio Menchero et al. |
CellBio 2022 – An ASCB/EMBO Meeting
This preLists features preprints that were discussed and presented during the CellBio 2022 meeting in Washington, DC in December 2022.
| List by | Nadja Hümpfer et al. |
EMBL Synthetic Morphogenesis: From Gene Circuits to Tissue Architecture (2021)
A list of preprints mentioned at the #EESmorphoG virtual meeting in 2021.
| List by | Alex Eve |
FENS 2020
A collection of preprints presented during the virtual meeting of the Federation of European Neuroscience Societies (FENS) in 2020
| List by | Ana Dorrego-Rivas |
ECFG15 – Fungal biology
Preprints presented at 15th European Conference on Fungal Genetics 17-20 February 2020 Rome
| List by | Hiral Shah |
ASCB EMBO Annual Meeting 2019
A collection of preprints presented at the 2019 ASCB EMBO Meeting in Washington, DC (December 7-11)
| List by | Madhuja Samaddar et al. |
Lung Disease and Regeneration
This preprint list compiles highlights from the field of lung biology.
| List by | Rob Hynds |
MitoList
This list of preprints is focused on work expanding our knowledge on mitochondria in any organism, tissue or cell type, from the normal biology to the pathology.
| List by | Sandra Franco Iborra |











 (No Ratings Yet)
(No Ratings Yet)